Welcome to A Traveller’s Guide to Feathers. Visit here for weekly updates on the most exciting recent discoveries from the world of bird biology. Your adventure begins below!
A Traveller’s Guide to Feathers, Article 108 -
Every Bird in the World
18 November 2018
We share the planet with more than ten thousand species of birds. An attempt to see representatives of every species would be a fool’s errand. Some birds live in spots so remote, and in such low numbers, that they haven’t been seen in many years. New species are discovered quite regularly, making “all” a vanishing target. Many enthusiasts turn to the pages of field guides and handbooks, allowing the descriptions and illustrations to fuel their imagination of birds that they are unlikely ever to see.
I did just that this week. I dived into my extensive ornithological library is search of birds that I had not stumbled across before. I skipped over some rather uninspiring birds, including the Brown Tanager and the Drab Seedeater, searching for beautiful creatures with exotic names. Here is what I found.

Painting of a Merida Flowerpecker by Joseph Smit – Proceedings of the Zoological Society of London (vol. 1870, plate XLVI)
The Merida Flowerpecker was discovered in 1871. It can be found in the scrubby highlands of the Mérida region of Venezuela. A teeny songbird, it is sometimes displaced from good foraging spots by hummingbirds. Like hummingbirds, this flowerpecker feeds mainly on nectar and small insects. Both males and females are mostly black or blueish-grey on top, with reddish-brown undersides. The genus name, Diglossa implies that the bird has two tongues or has two voices, while the species name, gloriosa, is the Latin form of the word glorious. Even though we know almost nothing about the breeding biology of this species, the International Union for the Conservation of Nature considers the Merida Flowerpecker to be of least concern.
The scholarly community has known about the Red-collared Widowbird since the late 18th century. This bird has a wide, but patchy distribution across the southern half of Africa, and it is known to occupy a wide range of habitats. Roughly twice the size of a flowerpecker, female Red-collared Widowbirds are nondescript in their colouration. Males could scarcely be more impressive. Some are all black, but most have a bright red slash across their throat. Most impressive is the male’s black tail, which is longer than the rest of the bird in some subspecies. Males with the longest tails attract two or three mates, while more poorly-ornamented males remain unmated. Males and females both contribute to nest building, but incubation and feeding of nestlings are carried out by the females alone. The genus name, Euplectes, refers to their woven nest. The species name, ardens, is Latin for glowing or burning, likely a reference to the males’ bright red collar.
The Snow Mountain Mannikin was unknown to the bird world until 1939, when it was found at an elevation of more than four thousand metres in the Snow Mountains of west-central New Guinea. It is a perfectly respectable little songbird with a black face, light brown bib, darker brown back, striped sides, and a robust grey bill suitable for cracking small seeds. The genus name, Lonchura, refers to the pointed tail feathers of some members of the group. What really stands out about this bird is how little we know about it. The IUCN lists the Snow Mountain Mannikin as having a stable population, but that is something of a guess. We do not know if it resides on its breeding range year-round, or if it wanders widely. The nest is built of grass, but nothing else is known of its breeding biology.
And that is one of the most wonderful things about bird biology. Although we know a great deal about birds, much awaits our investigations.
Photo credits: Painting of a Merida Flowerpecker by Joseph Smit – Proceedings of the Zoological Society of London (vol. 1870, plate XLVI) – https://de.wikipedia.org/wiki/M%C3%A9ridahakenschnabel#/media/File:DiglossaGloriosaSmit.jpg; Birds of Congo stamp collection, including a male Red-collared Widowbird – www.ebay.com; Snow Mountain Mannikin – www.pinterest.com
A Traveller’s Guide to Feathers, Article 107:
Now That We Understand the Problem…
11 November 2018
New Zealand Rockwrens are tough little birds. Living in high-altitude alpine habitat of New Zealand, their nests consist of excavated cavities within vegetated ledges, and cracks on cliff faces and boulders. At forty-four days, their nesting period is surprisingly long for a bird of that size. When confronted by a snowstorm during the breeding season, a parent may tunnel down through the snow to reach its chicks. Despite being warriors, could it be that New Zealand Rockwrens are helpless in the face of invasive mammalian predators?
If a plant or animals has been moved by humans from its native lands, across a natural barrier, and has managed to establish a self-sustaining population in that new spot, then it is considered to be “introduced.” If that species goes on to have negative consequences for the wildlife or landscape of its new home, or on human health or the economy, then the creature is considered to be “invasive.” A number of invasive mammals are causing trouble in New Zealand. These include stoats, house mice, rats and possums. Numbers of the New Zealand Rockwren have declined to the point where they are considered endangered. Are invasive, predatory mammals to blame?
Conservation biology Kerry Weston of New Zealand’s Department of Conservation and her colleagues studied the breeding biology of the New Zealand Rockwren, with particular emphasis on the potential impact of invasive mammals. At three high-altitude sites, Weston et al. searched for rockwren nests, and followed their progress. In some cases, nests were monitored using continuous video-recording and motion-activated cameras. Mammals were trapped and disposed of in some locales.
Weston and her crew located 146 rockwren nests, and reported on some interesting aspects of the birds’ breeding biology. For instance, both parents contributed to incubating the eggs and providing for the nestlings during the day, but females alone incubated eggs and brooded young at night. As the nestling grew in size, they received food every three or four minutes during daylight hours. Nestlings are noisy, and can sometimes be seen begging from the nest’s entrance.
The most common cause of rockwren reproductive failure was predation by stoats, rats and mice. Cameras revealed that stoats ate eggs, nestlings and brooding adults. However, in spots where predators were controlled, no predation by stoats was confirmed, and the leading cause of failure was nest abandonment, sometimes the result of extreme weather.
In their five-year study, just thirty-three stoats, ten rats and one mouse were captured. Despite the low number of mammalian predators in the landscape, it was clear that these animals had considerable negative consequences for populations of the endangered rockwren. The impact of invasive predators on insular and lowland bird populations is well established. Less well known is the effect of invasive mammals on birds breeding at high altitudes. Weston et al. wrote: “Novel predator control techniques that can be safely applied at scale above treeline are urgently required.” Let’s hope that such control will be forthcoming, and that the New Zealand Rockwren’s endanagered status will soon be changed.
Weston, K. A., C. F. J. O’Donnell, P. van Dam-Bates and J. M. Monks. 2018. Control of invasive predators improves breeding success of an endangered alpine passerine. Ibis 160:892-899.
Photo credits: New Zealand Rockwren stamp – www.pinterest.com; photograph courtesy of Pete McGregor - http://stmedia.co.nz/wgl-nzac/2011/02/have-you-seen-the-alpine-rock-wren/
A Traveller’s Guide to Feathers, Article 106
With This Ring…
28 October 2018
It is hard to imagine that the field of astronomy would have made much headway if telescopes had never been invented, or that cell biology would have advanced far without microscopes. Even though the most powerful tool in any scientific endeavor is a curious mind, the application of technology helps too.
Those techniques do not always have to be sophisticated or expensive. In a marvelous example of understatement, Sanjo Rose and Dieter Oschadleus of the University of Cape Town in South Africa recently wrote: “Bird ringing provides novel insights into the private lives of birds.” Let me be a little less restrained; without the application of numbered bands to the legs of millions of wild birds, much of the lives of birds would still be a mystery to us.
Bird ringing, known is some parts of the world as bird banding, and the subsequent recapture of some of those banded individuals, allows us to learn about where, when and how they live their lives. Ringed on its breeding territory, a bird might be recaptured on its wintering grounds, providing us with a link between the two regions. If a combination of coloured legs bands are used for individual identification, we can sometimes follow a bird as it interacts with its environment and other birds around it.
It is almost certain that some readers of this column will be engaged in bird ringing as a hobby. I have met hobbyists that have ringed tens of thousands of birds. I have ringed a far smaller number of birds for research purposes. All of this activity contributes to a better understanding of the avian world. In southern Africa, the activities of bird ringers is coordinated by the South African Bird Ringing scheme. Since 1948, more than 2.3 million birds have had numbered rings applied to their legs.
In a recent publication, Rose and Oschadleus reported on their efforts to learn about the longevity of estrildids, or waxbill finches, based on the banding and recovery of birds in southern Africa. Old World birds, estrildids can be found over large parts of Africa, southern Asia and Australasia. Within the group’s 130 species, some will be very familiar to fanciers of cage birds. These include the Australian Zebra Finch, Java Sparrow and Gouldian Finch. Rose and Oschadeleus found that more than 150,000 estrildids have been banded in southern Africa. Five species were represented by more than 10,000 ringing records each, including the Blue Waxbill and the Red-headed Finch. Many of those ringed birds have been recovered. Other species, including the Cinderella Waxbill and the Locust Finch have been ringed fewer than one hundred times, and none have been seen subsequently.
Longevity is a crucial demographic if we are to understand the population dynamics of species, and so Rose and Oschadleus reported those figures for southern African birds with the largest datasets. They found, for instance, that 27,813 Red-headed Finches had been marked, and that the oldest individual had been at least 5.2 years of age. More than twenty-thousand Bronze Manakins had received rings, but the oldest had been only 3.7 years of age. The record holder was a Blue Waxbill, one of 28,037 ringed. It had first been banded on June 25, 1996 and recovered on February 21, 2009. By my calculations, this bird’s minimum age was twelve years, seven months and twenty-eight days.
If you are involved in bird ringing, well done! You are contributing to a better understanding of birds. If you are looking for a new hobby, perhaps you will consider contacting the wild bird enthusiasts’ group in your area to find out how you could become part of the program.
Rose, S. and H. D. Oschadleus. 2018. Longevity summary from 69 years of Estrildidae ringing data in southern Africa. African Zoology 53:41-46.
Photo credits; Banding a Gouldian Finch fledgeling - ladygouldianfinch.com/features_banding.php; Cinderella Waxbill stamp - colnect.com
A Traveller’s Guide to Feathers, Article 105
Hippy Birds
21 October 2018
Smooth-billed Anis are the late-1960s, commune-dwelling hippies of the bird world. Found as far north as the southern United States, and residing over much of the Caribbean and Central and South America, as many as six females lay their eggs in a shared nest. Ani chicks are raised by the group, with the help of multiple males and non-breeding helpers. It all sounds like a synergistic, happy-go-lucky love fest.
Closer examination shows that the situation isn’t quite so lovely as it appears. Communal they may be, but members of a Smooth-billed Ani group sometimes bury eggs, or roll eggs up the side of the nest and toss them out. There are at least two functions for this ovicidal behaviour. By destroying the eggs of others, a female can reduce the competition for limited resources for her own chicks. If she buries older eggs, or tosses older eggs from the nest, she helps to ensure that her chicks will not be the youngest in the nest. In doing so, her chicks will not be at a disadvantage because of competition with older chicks.
Leanne Grieves and James Quinn of McMaster University in Canada studied the ovicidal behaviour of Smooth-billed Anis in Puerto Rico. They explained that when they are first laid, ani eggs are white because they are covered with a thin chalky layer. When that layer wears away over a period of days, the underlying blue colour of the shell is exposed. Grieves and Quinn asked whether female anis might use the blue colouration of shells to indicate that the eggs had been laid some days earlier, and use that cue to eject or bury eggs when laying their own.
The researchers scraped some eggs with a small brush to wipe away the chalky covering, leaving them blue. In order to introduce a level of control, other eggs were handled, but the chalky covering was not removed, and so the eggs remained white.
Grieves and Quinn found that blue eggs were no more likely to be buried that white eggs. Blue eggs were also no more likely to be thrown from the nest. Colour does not seem to be a cue. However, they found that females in larger communal groups were more likely to engage in both of the destructive behaviours than females in smaller groups. In larger groups with four or five females, an egg was twice as likely to be destroyed as an egg in a group with just two or three females.
Given their size, female Smooth-billed Anis lay very large eggs; nearly 18% of their body weight goes into each one. An egg is a large investment. The greater the group size, the greater the competition. In order to make her investment of energy and resources worthwhile, it seems that females in larger groups will go to great lengths to ensure that her eggs, and therefore her chicks, are the ones to survive.
Life can be tough, even in a commune.
Grieves, L. A., and J. S. Quinn. 2018. Group size, but not manipulated whole-clutch egg color, contributes to ovicide in joint-nesting Smooth-billed Anis. Wilson Journal of Ornithology 130:479-484.
Photo credits: Smooth-billed Ani – www.pinterest.com; Smooth-billed Ani stamp - http://www.birdtheme.org/mainlyimages/index.php?spec=498
A Traveller’s Guide to Feathers, Article 104
From the Wild, to Captivity, and Back
14 October 2018
Specialists in the field of biological conservation need to be simultaneously conservative and creative in their practices. When it comes to endangered creatures, we can rarely afford to be cavalier in using new, unproven methodologies. Even so, a technique that serve the needs of one threatened species may be entirely inappropriate for a different species. How can we be cautious and innovative at the same time?
It will be desirable to release animals to the wild after a period in captivity in several circumstances. For instance, individuals born in captivity, but released to the wild, may represent salvation for some species. Animals that have been confiscated by authorities may be used to bolster a declining wild population. It may be deemed useful to translocate individuals from a healthy wild population to an area from which the species has been eliminated.
Consider the Spix’s Macaw, native to northeastern Brazil. According to the IUCN, this Macaw is critically-endangered, and possibly extinct in the wild. With luck, the few individuals held in captivity will allow us to reestablish wild populations in the future. However, we don’t currently know the best way to ensure this. How do we obtain answers without risking the few individuals that are left?
Alice Lopes of the Instituto de Pesquisa e Conservaçăo in Brazil and her colleagues studied Blue-fronted Amazons that had been released to the wild after a period in captivity. The IUCN lists this parrot as of least concern. Lessons learned from releases of this reasonably common parrot might be applied to other, more-threatened species. The goals of Lopes and her colleagues was to “evaluate techniques of management and monitoring employed in the translocation of a group of captive-raised Blue-fronted Amazons.”
Thirty-one Blue-fronted Amazons were obtained from the Wild Animal Triage Centres (CETAS) in Brazil. Each had been in captivity for at least three years. These birds were studied at a site at a private farm in southeastern Brazil. Before release, the birds were trained to recognize predators. They were held in large aviaries that allowed them to practice the skills they would need to survive in the wild. After ten months of training, the parrots were permitted to leave, although they were permitted to return to the aviaries, and were provided with food to supplement what they found in the wild.
The thirty-one parrots released had a range of fates. Three were known to have perished. The whereabouts of five others could not be determined. Ten others showed behaviours that suggested they were adapting to life in the wild. The remaining thirteen individuals continued to behave in a way that better suited captive life, such as remaining near the aviary, and interacting with humans. It appeared that some members of the final group were captured by people in the area.
The study of Lopes et al. showed that “confiscated ex-pet Blue-fronted Amazon Parrots can be good candidates for translocation, but a training program should be applied to them prior to their release.” For instance, subjects need to learn to stay away from humans in order to avoid being recaptured.
Wild populations of the Blue-fronted Amazon range from Bolivia to northern Argentina. Even though it is considered to be of least concern, its numbers in the wild are in decline. According to the IUCN, between 1981 and 2005, 413,000 wild-caught individuals are known to have been sold internationally. Between 2008 and 2010, the CETAS of Brazil received 3,395 individual Blue-fronted Amazons, of which 2145 were seized. A quick search of YouTube will reveal why the Blue-fronted Amazon is so popular as a cage bird. It is to be hoped that will remain sufficiently common in the wild so as to continue to serve as a model for studies of conservation practices.
Lopes, A. R. S., et al. 2018. Translocation and post-release monitoring of captive-raised Blue-fronted Amazons Amazona aestiva. Acta Ornithologica 53:37-46.
Photo credits: Blue-fronted Amazon – fotolia.com & clipground.com
A Traveller’s Guide to Feathers, Article 103:
Naughty Bird Biologists
7 October 2018
The Wilson Journal of Ornithology is a highly respected repository of information related to bird science. The most recent issue of the journal includes sixteen long articles and twelve briefer communiques. Readers of that issue can learn about research on the effects of flooding of the Missouri River on Least Tern reproduction, the nesting behaviour of Gray Tinamous in Ecuador, and parental behaviour of Costa Rican Clay-coloured Thrushes.
The journal began one-hundred and thirty years ago under the name The Ornithologists’ and Oologists’ Semi-annual. That title lasted only a year, being replaced by the more ponderous Semi-annual, Agassiz Association, Department of the Wilson Chapter. The name was changed to The Wilson Quarterly in 1892, and then The Journal of the Wilson Ornithological Chapter of the Agassiz Association in 1893. Things settled down after that, and the journal was called The Wilson Bulletin from 1894 to 2005.
In the early years, most subscribers to the journal were dedicated laypeople, rather than professional ornithologists. Articles from more than a century ago reflected the naive state of the field at the time. An account of birds seen while on a cruise might have been followed by an description of the timing of nesting of birds in a local cemetery. Even so those old issues can for interesting reading.
The second issue of the fifth volume of the journal was just eight pages long. On the second page of that issue was a statement short on details but rich in inuendo. It read: “Charges of sufficient seriousness have been preferred against Messrs. J. W. P. Smithwick and F. T. Pember to warrant their expulsion from the Chapter. A majority of the Executive Council have voted to remove their names from our roll. Their crime is fraudulent methods in Oology.”
It seems that J. W. P. Smithwick of San Souci, NC, and F. T. Pember of Granville, NY, had been active members of the group to that point, including writing articles with titles like: “Collecting in the Gila Valley” and “The Burrowing Owl: Speotyto cunicularia hypogaea.” Despite being on the membership list in Volume 5, issue 1, their names were missing from that list in issue 2.
I have not discovered what crimes against oology had Smithwick and Pember dismissed from the society, but I have found a few additional details about their comings and goings. Franklin Tanner Pember was born in 1841 and died in 1924. A businessman with interests in oil and oranges, his boyhood fascination with nature led to a substantial collection of artifacts, including bird nests and eggs. He and his wife, Ellen Wood Pember, established the Pember Library and Museum in 1909. That facility still serves the people of eastern New York state.
An online article by David Cecelski suggests that John Washington Pearce Smithwick was born in North Carolina in 1870, and was an avid collector of bird eggs by 1888. Shortly after being drummed out of the Wilson Society, Smithwick donated his collection of eggs to the state’s natural history museum. He then became a physician. I have not seen Cecelski’s reference material, and so cannot comment on his assertion that Smithwick was a hate-mongering racist.
Birds are angelic. Birds biologists will be judged by time.
Photo credits: Photo credits: reprint of The Ornithologists’ and Oologists’ Semi-annual Volume 1 (1889) – www.empik.com; image from page 61 of “The Ornithologists’ and oologists’ semi-annual” (1889) - https://www.flickr.com/photos/internetarchivebookimages/14750528982/
 A Traveller’s Guide to Feathers, Article 102 -
A Traveller’s Guide to Feathers, Article 102 -
The Early Bird
30 September 2018
Birds that migrate to far-northern latitudes for breeding may arrive to find a wealth of opportunity. Grazing geese are likely to find endless fields of fresh grass. Insect-eating pipits and wagtails may find their protein-rich quarry to be a non-depletable resource.
Bird biologists have found that the first individuals to return from their wintering areas to their northern breeding grounds generally have greater reproductive success. The causes of this success are many. The first birds back may have the greatest opportunity to recover from the challenges of migration, and could find access to the best breeding territories. If the first breeding attempt fails, early breeders have a greater opportunity to try again. Perhaps earlier males can increase their reproductive success by mating with females who are not their social mate. By breeding earlier, their chicks may have a longer period to grow and acquire the reserves necessary for successful southward migration.
It is possible, of course, that some birds will migrate northward too early, and will arrive to find their breeding grounds in the grip late-Winter weather. Even so, on the whole, earlier seems to be better.
However, as with so many aspects of bird biology, this phenomenon has been reasonably-well studied in species living in temperate zones, with far less information available for tropical species. Vanessa Bejarano and Alex Jahn of the Universidade Estadual Paulista in São Paulo, Brazil, sought to address that gap in our knowledge.
Between 2013 and 2016, Bejarano and Jahn studied a population of Fork-tailed Flycatchers in the Estação Ecologica de Itirapina in southern Brazil. Described as intra-tropical migrants, these flycatchers come to their breeding grounds from their off-season homes in northern South America. The birds were colour-banded for individual identification. The researchers monitored flycatchers on the study site, noting when they first arrived, and followed their breeding activities.
Life in the tropics can be challenging for birds, and Bejarano and Jahn found that only ten percent of nesting attempts were successful. On average, male flycatchers were seen on the breeding grounds earlier than females, a phenomenon known as protandry. Males that arrived first attracted females that laid eggs earlier. Nest success was higher when egg laying was earlier. In summary – earlier is better. Although this relationship between dates of arrival on the breeding ground and breeding success has been established for a number of birds in temperate regions, this seems to be the first study to show the association for birds that live within the tropics.
Further study will be needed to determine how male Fork-tailed Flycatchers manage to arrive to breed before females do, and why earlier arrival translates into greater success. The authors suggested that: “further research on the subject would benefit from a multi-disciplinary, comparative approach among migratory systems.” In an era of global climate change, the results of such research is likely to prove valuable.
Bejarano, V., and A. E. Jahn. 2018. Relationship between arrival timing and breeding success of intra-tropical migratory Fork-tailed Flycatchers (Tyrannus savana). Journal of Field Ornithology 89:109-116.
Photo credits: Brasilian stamp – www.pinterest.com; painting by John James Aububon – www.amazon.com
A Traveller’s Guide to Feathers, Article 101 -
Endangered Birds on Refuse Sites
23 September 2018
As recently as 2009, the IUCN considered Africa’s Hooded Vulture sufficiently abundant as to be of “least concern.” Its status was downgraded to “endangered” in 2011, and dropped again in 2015 to “critically endangered,” and that is where this vulture sits today. It isn’t that Hooded Vultures are particularly hard to find; there are thought to be as many as 197,000 of them. All told, they have a comparatively large range in Africa, including many countries from Angola to Zimbabwe. Instead, the Hooded Vulture is considered to be critically endangered because of an extremely rapid decline in its abundance. According to the IUCN, this decline is the result of “indiscriminate poisoning, trade for traditional medicine, hunting, persecution and electrocution, as well as habitat loss and degradation.”
 The bird might be rare elsewhere, but in recent years it seemed to be doing quite well in Ghana, where it was seen commonly in landfill sites, food markets, urban dumpsters and around slaughterhouses. In Ghana’s capital city, Accra, flocks of five hundred vultures might be seen. Then, at least according to anecdotal evidence, Hooded Vultures started rapidly decreasing in number.
The bird might be rare elsewhere, but in recent years it seemed to be doing quite well in Ghana, where it was seen commonly in landfill sites, food markets, urban dumpsters and around slaughterhouses. In Ghana’s capital city, Accra, flocks of five hundred vultures might be seen. Then, at least according to anecdotal evidence, Hooded Vultures started rapidly decreasing in number.
Francis Gbogbo of the University of Ghana, Japheth Roberts of the Ghana Wildlife Society, and Vincent Awotwe-Pratt of the Conservation Alliance saw the need for a critical examination of the Hooded Vulture’s abundance in its urban stronghold in Ghana, and an investigation of its principle sources of mortality.
The study was conducted in Accra. From just 377,000 residents in 1960, the human population of the city has grown to as many as 4,000,000 in the greater metropolitan area. Weekly counts of Hooded Vultures were made on the Legon Campus of the University of Ghana. As many as 220 vultures were seen between November 2010 and January 2011, but this number plummeted to just five individuals in 2015 and 2016. The numbers seemed to represent an actual decline, and not just a relocation. Some of the decline in vulture numbers may have been a result of competition with Pied Crows which increase in number on the university campus over the same period. The introduction of covered waste bins on campus may also have decreased the opportunity for scavenging by vultures.
Similarly, in an attempt to decrease the risk of collisions between airplanes and vultures, efforts were made to decrease the attractiveness of the region around Kotoka International Airport. For instance, refuse dumps in the area were closed or covered. The recent closure of unhygienic slaughterhouses in Accra likely also had an affect on Hooded Vulture numbers.
The researchers examined the published literature for details of the vulture’s abundance. Gbogbo et al. also spoke with vulture researchers, as well as those who had experience with the habitats frequented by Hooded Vultures, including waste managers, scrap dealers and refuse scavengers. It seems that the capture and sale of Hooded Vulture carcasses for use in traditional medicines and black magic is not insubstantial. The researchers found that the value of the head of a vulture in a market in Tamale was 200 Ghana cedis. That is about two-months’ wages for a middle-income earner in Ghana. Many dead vultures and vulture parts are apparently exported to Nigeria. Some vultures are being captured and sold as food, sometimes to an unsuspecting public.
Nothing about conservation of the Hooded Vulture is likely to be simple in Ghana. Clearly it is not appropriate to reopen refuse dumps in order to provide foraging opportunities. Gbogbo and his coworkers identified “the need for an intensive education of the Ghanaian populace on the significant status and need for the protection of the species.”
Gbogbo, F., J. S. T. Roberts and V. Awotwe-Pratt. 2016. Some important observations on the populations of Hooded Vultures Necrosyrtes monachus in urban Ghana.
Photo credits: Hooded Vulture photograph – www.pinterest.com; Hooded Vulture stamp - http://www.wnsstamps.post
A Traveller’s Guide to Feathers, Article 99:
Hope
09 September 2018
In the previous installment of A Traveller’s Guide to Feathers, I described recent research on the natural history of the Okinawa Rail. Dr. Shun Kobayashi and his colleagues discovered, among other things, that this endangered rail is omnivorous, exploiting a wide range of plant and animal foods, and that they forage in more than one habitat.
Wanting to know more about the Okinawa Rail and other threatened wildlife in the area, I contacted Shun Kobayashi, a post-doctoral researcher at the University of the Ryukyus. He responded immediately with great energy and enthusiasm. He pointed out that the Okinawa Rail has become a symbol of Okinawan animal life. Consequently many people in the region are aware of the bird, but many of those are unaware of its plight. Predation by introduced mongooses and cats is likely the most serious source of rail mortality, but habitat loss and collisions with automobiles also contribute. The population fell through 2007, before beginning to recover.
Shun Kobayashi told me that the Ministry of the Environment became involved, creating a breeding program, and attempting to remove mongooses from northern parts of the island where their impact on native wildlife is greatest. Consequently, rails are becoming more numerous, although some threats remain.
Under the guidance of Professor Masako Izawa, university students continue to consider the natural history of Okinawa Rails. One student tracks individuals in the wild, and another studies captive individuals. The professor’s website describes other work that her group is engaged in. “We conduct research on mammal home ranges, environmental utilization, social structure, breeding behavior, feeding habits… Animals that students have researched… include the Iriomote wild cat, the Tsushina leopard cat, the flying-fox, Cervus nippon keramae, different types of mice, the blue rock thrush, and more.” The University of the Ryukyus has a very navigable website: http://www.u-ryukyu.ac.jp/en/.
Shun Kobayashi pointed out that Dr. Kiyoaki Ozaki, a researcher at the Yamashina Institute for Ornithology, has been studying Okinawa Rails for more than thirty years. You can learn more about the Yamashina Institute at: http://www.yamashina.or.jp/hp/english/index.html.
Shun Kobayashi explained that his interests were not just for our avian friends, but also for plant pollination systems that depend on mammals, and the ecology of animals that are found only in the Ryukyu Islands. He is, for instance, currently studying the impact of dogs and cats on the native animals of Okinawa-jima Island. I was told that the support of non-governmental organizations, such as the Conservation and Animal Welfare Trust, Okinawa, are key elements in the study of wildlife in the area. “I could not do the research without their support,” wrote Shun.
Despair only occurs when no hope remains. I see no need for despair for our natural world as long as people such as Dr. Shun Kobayashi, Professor Masako Izawa, and Dr. Kiyoaki Ozaki believe that there is a future.
Photo credits: Okinawa Rail successfully crossing a road – www.pinterest.com; map showing the location of Okinawa Rails – Yamashina Institute for Ornithology (http://www.yamashina.or.jp/hp/english/okinawarail/about_or.html)
A Traveller’s Guide to Feathers, Article 98:
A Rail with a Future
02 September 2018
Recently I was discussing the Invisible Rail with a colleague. It is a rarely-seen bird that was first collected by Alfred Russel Wallace in the late 1800s. It is found on only the island of Halmahera in the North Moluccas of Indonesia. I explained to my colleague that the discovery of the Invisible Rail on a second island was something of a mystery given that the bird is presumably flightless.
“What makes you think that the Invisible Rail is flightless?” he asked. As a group, rails have a remarkable inclination to arrive on islands, loose their ability to fly, and then evolve into a form that is distinctive from other rails. If they have come to inhabit an island with no ground-dwelling predators, and lots of food on the ground, then flight is rather a waste of precious energy.
Since flightless rails on small islands often have small populations, and because recently-introduced predators are a common feature of islands, many rail species are under threat, and others have already fallen to extinction. Because so few ornithologists live on tiny islands, flightless rails are also a poorly-known group.
One of the very first requirements of a successful conservation program is a thorough understanding of the natural history of the threatened species. To this end, Shun Kobayashi of the University of the Ryukyus and twelve of his colleagues studied the diet of the endangered, almost entirely flightless Okinawa Rail. This bird is found only on the northern part of Okinawajima Island of Japan.
The IUCN’s description of the plight of the Okinawa Rail is not particularly optimistic. “This species is listed as Endangered because it has a single, very small population within a very small range on just one island, and both its range and population are undergoing continuing declines as a result of introduced predators and the loss of forest to logging, infrastructure development, agriculture and construction of golf courses.”
A through study of the diet of an endangered, ground-dwelling forest bird is not necessarily easy. Direct observation are likely to be hampered by the undergrowth, and sacrificing individuals to examine their gut contents is clearly not an option. Instead, Kobayashi and his co-workers examined the contents of the gizzards of 189 Okinawa Rails that had been killed by introduced Indian mongooses or traffic accidents.
The gizzards examined revealed a very large range of animals eaten, as well as eighteen species of plants. Among animal prey, four species of snails were particularly common. Insects, mainly beetles, grasshoppers and ants, also contributed greatly to the diet of these birds. The researchers did not hesitate to describe the Okinawa Rail as “omnivorous.” The study made it clear that these rails forage in more than one habitat type, as their gizzards contained millipedes from both open fields and forest edges, as well as hermit crabs that live on the coast. The abundance of land snails in the Okinawa Rail’s diet distinguishes it from other rails.
If Kobayashi and his colleagues had discovered that the Okinawa Rail had a particularly narrow diet, eating only the flowers of a very rare plant for instance, the bird’s long-term prospects might be less favorable. Finding that the rail is capable of exploiting a wide range of food types is one step forward in efforts to ensure the bird’s long term survival. In the next installment of A Traveller’s Guide to Feathers, you will find more about the wonderful work being done to conserve Japanese wildlife.
Kobayashi, S., et al. 2018. Dietary habits of the endangered Okinawa Rail. Ornithological Science 17:19-35.
Photo credits: Okinawa Rail stamp – ww.lelong.com.my; art - amanaimages.com
A Traveller’s Guide to Feathers, Article 97:
Greenlanders Can Breathe More Easily
19 August 2018
I read recently that there are more chickens than there are birds of all other 10,000 species of birds combined. That is a lot of chickens. I suppose it explains why virtually every article I find in scholarly journals relating to disease and pathology in birds concerns chickens. Of the very few studies that do not consider chickens, the vast majority are about turkeys. I was delighted to find a recent study of avian diseases that was about wild birds.
There is no doubt that the viruses responsible for avian influenza are a major concern. They have the potential to devastate poultry production (again, chickens!), and represent a serious potential threat to human health. Wild birds may play a role in the transmission and dispersion of avian influenza viruses to chickens and human populations, and may provide an opportunity for the evolution of viral agents. Anything that we can learn about avian influenza in wild birds may help us to combat future outbreaks.
Migrating birds are a special concern. Infected individuals might move diseases over vast distances. Luckily, birds migrating in the world’s Eastern and Western hemispheres tend to remain separate, limiting their potential to cause a global epidemic. There is, however, an exception.
There are spots in the high Arctic where birds that winter in different hemispheres come together to breed in very large numbers. Northeastern Greenland is one such place. Tens of thousands of breeding Barnacle and Pink-footed Geese congregate in this region, where they are joined by migratory gulls, terns, waders and ducks of at least eighteen species. These birds have the potential to spread avian influenza viruses between Europe, Africa and North America. Furthermore, it has been suggested that in the cold of the far north, viruses excreted by breeding birds one year may survive to infect birds the following year.
French researcher Nicolas Gaidet and his colleagues visited Greenland’s Jameson Land, which they described as “a vast tundra area with scattered lakes.” They collected three hundred samples of fresh goose feces, and 28 feather samples from geese that were found in large flocks. Water samples were also taken from lakes that were home to breeding geese. Those samples were subjected to genetic analysis designed to detect avian influenza viruses.
The results of a study can rarely be summarized in a single sentence, but they were in the case of the study of Gaidet et al. “No AIV was detected in any fecal, feather or water sample tested.” Despite the high density of birds in the area providing an opportunity for transmission of avian influenza viruses, no evidence of it was found. This is, of course, good news. Particularly for chickens. Even so, the researchers pointed out that Greenland, as a crossroads for birds of different hemispheres, “warrants further surveillance for AIV in wild birds breeding or staging to better understand the role of high Arctic regions for intercontinental movements of AIVs.”
Gaidet, N., I. Leclercq, C. Batéjat, Q. Grassin, T. Daufresne, and J.-C. Manuguerra. 2018. Avian influenza virus surveillance in High Arctic breeding geese, Greenland. Avian Diseases 62:237-240.
Photo credits: Barnacle Goose – roaringwaterjournal.files.wordpress.com; Pink-footed Goose – www.pinterest.com
A Traveller’s Guide to Feathers, Article 96
House Sparrows in Kentucy
12 August 2018
The Wilson Journal of Ornithology is a respected repository of all matters pertaining to bird biology. In order to collect data worthy of publication in that journal, a researcher might have to spend years in crocodile-infested swamps or leech-filled jungles. The most recent volume of the publication includes articles on the breeding biology of Gray Tinamous in Ecuador, ovicide in Smooth-billed Anis in Puerto Rico, and assortative mating by Plain Laughingthrushes in China.
However, reports in the journal were not always so exotic. The original name of the Wilson Journal of Ornithology was the Ornithologists’ and Oologists’ Semi-annual. The very first issue was published in 1890. On pages 11 and 12 of that issue was an article entitled “Observations from the Deck of a Steamer” by Otley Pindar. A surgeon by profession, it seems that Pindar took a journey on the steamboat Granite State in the summer of 1889, embarking from Hickman, Kentucky. He sailed along both the Mississippi and Ohio rivers, spending his time on the ship’s upper deck watching birds, rather than sipping bourbon in the saloon.
Pindar provided readers of the O & O Semi-annual with his thoughts on seventeen species of birds he saw. Forester’s Terns, Chimney Swifts and Barn Swallows skimmed and darted near the boat, while Turkey Vultures circled high overhead. Downstream from Columbus, he spied a family of “Kingbirds,” which were almost certainly the species known today as the Eastern Kingbird. On the upstream side of Columbus, Pindar found Kildeers and “Snowy Herons,” which seems to be an old name for Snowy Egrets.
The dense swamps of Missouri provided roosting sites for American Crows, and Pindar spied what I suspect to be Fish Crows in both Shawneetown, Ill, and Lewisport, Ky. He encountered Great Blue Herons frequently as they flew across the river ahead of the boat. White-bellied Swallows, now known as Tree Swallows, entertained Pindar with their “aerial evolutions.” Riverside fields and forests were home to Common Grackles, American Robins, Eastern Meadowlarks and Field Sparrows. Henderson, Ky, gave Pindar the opportunity to see a very large colony of Bank Swallows and a covey of Northern Bobwhites.
Pindar was not able to identify every bird he saw, but those that he did seemed to have made his voyage all the more pleasant. Only one species of bird drew his ire. He wrote about the House Sparrow, an introduced species, describing it as “one which could be well dispensed with… found in abundance in every place at which the boat stopped, especially the larger towns and cities.”
In 1923, Pindar became the first ever president of the Kentucky Ornithological Society. He came upon this honor, in part, because he was judged to be the oldest practicing ornithologist in the state. The society is still around; its Fall 2018 meeting will be held the weekend of 14 to 16 September at the Pine Mountain State Resort Park. The House Sparrow is still around too, and can still be found in towns and cities. Pindar is long gone.
Pindar, L. O. 1890. Observations from the deck of a steamer. The Ornithologists’ and Oologists’ Semi-annual. 1:11-12.
Picture credits – stamp – www.birdtheme.org; photograph – www.pinterest.com
Traveller’s Guide to Feathers, Article 95
Bald Eagles Aren’t Really Bald
05 August 2018
One of the greatest joys of being involved with the study of birds for a very long time is the opportunity to watch explanatory ideas unfold. An hypothesis that is accepted by one generation of bird biologists can be overturned completely by the next. For instance, when I was a university student, I learned that birds such as vultures and ibis had bald heads because it helped them to stay clean. If you spend your day with your head in garbage, or blood-soaked corpses, it could be challenging to keep your feathers in good condition.
It seems that that story is more complicated than that…
Birds with dark plumage probably benefit when their feathers absorb solar radiation. Darkly-coloured individuals likely spend less energy keeping warm than they would if they were lighter in colour. That is not the only potential advantage of dark feathers, but it seems to be one of the most important ones. However, at some times of the year, or at some times of the day, the heat absorbed by dark plumage might become too much of a good thing. Could it be that the bald heads of some darkly-coloured birds serve as radiators of excess heat?
Ismael Galván and his Spanish colleagues at the Doñana Biological Station in Seville and the University of Córdoba addressed the possibility that some birds are bald because it allows them to radiate heat when conditions get to warm. The group studied adult Northern Bald Ibises at Zoobotánico Jerez, a zoo and botanical garden in southern Spain. The body of this bird is covered completely in black feathers, except for a bald head which is red. The intensity of this red colouration can change rapidly when more blood is diverted to vessels close to the skin’s surface. The blood in these vessels would bring heat from the core of the body to the surface. All of this suggests that individuals might be able to use their heads as radiators to prevent overheating.
The study was conducted in July, the warmest part of the year in that region. Galván et al. collected thermal images of semi-captive ibises over a wide range of environmental temperatures. Over a range of conditions, it was possible to determine the temperature of the feathered parts of the body, as well as the temperature of the naked head. As these measurements were being made, the group also quantified the intensity of red colouration of each subject’s head.
The warmer the day, the warmer the bird. The warmer the bird’s head, the redder it was. “Norther Bald Ibises lost considerable heat amounts through the unfeathered head,” wrote Galván and crew. This radiation of heat continued until the environmental temperature rose to 35º C, about 95º F, after which the radiator was no longer effective. The researchers are not currently able to say whether the increased flow to the skin of the head when the birds warms up is the result of a simple physiological response, or whether the bird is able to control the radiator.
This line of inquiry might be extended to include other species of bald birds, including the Bald Starling (aka Coleto), the Bornean Bristlehead, the Bald (Noisy) Friarbird, and the Bald Parrot. However, the study would be pointless on Bald Eagles, because…
Galván, I., D. Palacios and J. J. Negro. 2017. The bare head of the Northern Bald Ibis (Geronticus eremita) fulfills a thermoregulatory function. Frontiers in Zoology 14:15.
Photo credits: www.pinterest.com
A Traveller’s Guide to Feathers, Article 94
A Sinking Feeling
29 July 2018
Some numbers are so large as to be almost impossible to comprehend. For instance, China’s farmlands occupy 1.3 million square kilometres, more than half a million square miles. To put that value in perspective, it is more than two-and-a-half times the land area of Spain. Lots of mouths to feed require lots of land for crops. All of that farmland provides for the needs of some birds, but not others.
The story of the exploitation of resources becomes a bit more complicated when we read that 40% of farmlands in China are underlain by deposits of coal, a precious commodity. The extraction of coal in China has resulted in subsidence of land on a very large scale. This subsidence has required the abandonment of many rural agricultural villages, and relocation of the people who lived there.
Chunlin Li and colleagues at Anhui University and the Ministry of Environmental Protection studied the impact of land subsidence and the abandonment of rural villages in areas with coal mining on the abundance and diversity of birds. They conducted their study in the Huaibei Plain in the North China Plain, an region important for both farming and coal mining. In this region, the area affected by subsidence was growing at a rate of more than twenty square kilometres each year. When villages were abandoned because of subsidence, they were generally left as they had been.
Li et al. compared birdlife in inhabited villages and abandoned villages, counting birds that were seen or heard. The researchers were interested in both bird abundance and diversity, but also the structure of vegetation in the two habitat types. They found that abandoned villages had greater cover by shrubs and grasses, with more layers of vegetation. Field surveys documented more than 9,000 birds of 47 species. Abandoned villages were home to nearly twice as many birds and twice as many species as those villages that were still inhabited. Among birds that were more likely to be seen in abandoned villages were Common Hoopoes, Grey-capped Greenfinches, White Wagtails, and Long-tailed Shrikes.
The simplest messages might be that coal mining led to land subsidence, which led to abandonment of villages, which led to a greater abundance and diversity of birds. However, a curious thing happened to villages that had been abandoned because of subsidence. Abundant rainfall, combined with a high groundwater table, meant that after a few years, land that has collapsed because of coal mining turned into ponds, reservoirs and small lakes. Birds that had been taking advantage of abandoned villages would soon be replaced by wetland species. For some species, the opportunities provided by village abandonment would be short-lived.
Li,C., P. Cui, S. Zhou and S. Yang. 2018. How do farmland bird communities in rural settlements respond to human relocations with land subsidence induced by coal mining in China? Avian Conservation and Ecology 13(1):6.
Photo credits: Grey-capped Greenfinches, photograph by John Fish (right) and White Wagtail (bottom) – www.pinterest.com
A Traveller’s Guide to Feathers, Article 93
A Place Where Many Herons Come
20 May 2018
I like simple definitions. According to the Gage Dictionary of Canadian English, a heronry is: “a place where many herons come in the breeding season.” Perfect. It seems that Ramesh Roshnath and Palatty Alesh Sinu of the Central University of Kerala like definitions to be a little more comprehensive. To them, a heronry “is group nesting of colonial waterbirds” which might include herons, storks, pelicans, cormorants, ibises, and spoonbills, which cluster their nests in time and space.
Roshnath and Sinu had the right to claim that their definition is closer to the mark, as they have been studying the avian residents of heronries of the Kannur and Kasaragod regions of southwestern India. The waterbirds that nest in heronries in this region include Little and Indian cormorants, Little, Intermediate and Great egrets, Oriental Darters, and both Purple and Grey Herons.
We all recognize that the conversion of intact operating native habitat into urban settings is not going to stop. Does that conversion need to be completely devastating to native wildlife, or is sustainable development possible? Roshnath and Sinu set out to document the impact of urbanization on colonial waterbirds, with the hopes of finding ways to make the lives of these birds as productive as possible.
The researchers found fifty-two heronries in spots which included wild regions such as mangrove inlets, but also in trees adjacent to roadways, and in urban and residential sites. They visited the sites at the peak of the breeding season, counting and identifying birds’ nests, and recording details of the nesting trees such as species, height and diameter.
It was no surprise to find that some of the waterbirds nested more frequently in wetlands habitat than in cities. These birds included Purple Herons and Great Egrets. Curiously, some birds were more likely to engage in breeding in trees in city landscapes, including Black-crowned Night Herons and Little Cormorants. Over 1,500 nests were documented in trees along major roadways, but fewer than 500 in mangrove trees. Just 285 nests were seen in trees at forts and temples, and eighty in residential areas. The most popular trees for nesting were rain trees, which are highly valued as urban shade trees. Rain trees provide the birds with materials with which to construct their nests, and a branching patterns that facilitates nesting. Also popular with the birds were copper pod trees, jackfruit, mangoes, and banyan trees.
Beyond abundant suitable nesting trees for waterbirds, it seems that the urban environment in this part of India provides reduced predation pressure from snakes and birds of prey. Further, urban fish markets may provide waterbirds with part of their diet in the breeding season.
Some heronries in this part of India have likely been in use for a very long time. The continued use of some of these sites is not certain. Trees that are centuries old are felled to allow for the expansion of roads. Birds abandon heronries when bridges and fly-overs are built at the level of the tree canopy.
When it comes to sustainable development, simple solutions are often the best. “Posters regarding the nesting birds species could be posted under the trees to draw public attention and create awareness,” wrote Roshnath and Sinu. “Local governments may consider erecting heronry guards above the bus waiting shelters if nesting trees are located in such places.” If trees need to be felled for a city’s expansion, workers might selectively remove non-nesting trees first.
By these means might herons (and other waterbirds) continue to use traditional heronries.
Roshnath, R., and P. Allesh Sinu. 2017. Nesting tree characteristics of heronry birds of urban ecosystems in peninsular India: implications for habitat management. Current Zoology 63:599-605.
Photocredits: Oriental Darter and Purple Heron with chicks – www.pinterest.com
A Traveller’s Guide to Feathers, Article 92
The Single Best Solution
06 May 2018
When it comes to finding the single best solution to a challenge, I suspect that there are few challenges greater than those faced by parent birds. If the best solution is the one that will allow a parent to make the greatest possible genetic contribution to following generations, then the questions to be answered are nearly endless. Shall I attempt to mate monogamously, polygamously, or promiscuously? If I mate monogamously, how much of an effort should I make, and how much work shall I leave for my partner? Shall I put all of the year’s efforts into a single breeding effort, or shall I try to breed twice? Is it best to remain faithful to my partner, or to cheat? What is the best solution?
Japanese Tits are tiny songbirds that breed, not surprisingly, in Japan, as well as in surrounding parts of mainland eastern Asia. They are short-lived, and are socially monogamous. They lay their eggs and raise their nestlings in cavities, and will utilize nestboxes if given the opportunity. Many pairs will attempt to raise two broods in a year, rather than just one. Given that the average lifespan of a Japanese Tit is just two years, a second brood is likely to greatly increase an individual’s lifetime reproductive success.
Daisuke Nomi, Teru Yuta and Itsuro Koizumi of Hokkaido University and Niigata University in Japan studied the breeding behaviour of Japanese Tits in the Tomakomai Experimental Forest of Hokkaido for three years. The researchers were particularly interested in the effort that male tits made in provisioning their young. A male that makes a great effort to feed his chicks may realize greater reproductive success, but not if his partner responds by reducing the effort she makes. If a male makes a greater number of feeding trips this year, he may wear himself out and have a lesser probability of surviving to breed again next year.
Or… Could it be that a male that makes a particularly great effort in the year’s first breeding attempt will free his mate to recover from the stresses of breeding, and so increase the chances that she will be able to breed a second time?
Nomi and his colleagues utilized two Japanese Tit breeding sites, each provisioned with 150 wooden nestboxes. These boxes were checked frequently to document laying date and the number of eggs in each nest. Parents at each nest were marked with coloured and numbered leg bands. Both parents and their chicks were weighed, and the length of their legs measured. The rate at which the parents provided food to the chicks at each nest was determined using video cameras.
The greater the number of feeding trips made by the male, the greater the mass of the chicks in that nest. Beyond that, pairs in which the male made a high contribution were more likely to make an attempt to breed a second time in the same year. Ultimately, the decision about whether to make a second breeding attempt is very likely made by the female. Her decision seems to be made, in part, on the basis of the work ethic of her partner. “Paternal care may be more influential than previously though,” wrote Nomi et al.
Given the obvious advantage of making a large contribution to nestling care, why do not all males feed their chicks as fast as they possibly can? A male that spends all of its time and energy feeding chicks may be spending less time on essential defense of the breeding territory. If a male doesn’t give himself a break occasionally, he may diminish his chances of surviving until the following year. A male with a bit of free time may try to engage in copulations with females on neighbouring territories, and so spread his genes that way.
When it comes to birds and reproduction, the single best solution may not be as simple as it might seem.
Nomia, D., T. Yuta and I. Koizumi. 2018. Male feeding contribution facilitates multiple brooding in a biparental songbird. Ibis 160:293-300.
Photo credits: Japanese Tit stamp - www.birdtheme.org; Japanese Tit photograph – www.pinterest.com
A Traveller’s Guide to Feathers, Article 91
The North Wind Doth Blow
29 April 2018
Some bird species have an incredibly small distribution. Consider, for instance, the Helmeted Honeyeater. The state bird of Victoria, Australia, very small numbers of this bird can be found in the Yellingbo Nature Conservation Area, and almost nowhere else.
At the other end of the distribution scale are bird species with ranges that almost defy belief. One such bird is the Eurasian Tree Sparrow which can be found from Afghanistan to Brunei; from Christmas Island to Iraq; from Japan to Ireland. Beyond its native distribution, Eurasian Tree Sparrows have been successfully introduced to far-flung lands including Canada, southern Australia and the Philippines, and are sometimes seen in Israel, Gibraltar, and Timor Leste. These sparrows are clearly incredibly flexible and tolerant. However, the flexibility and tolerance of no creature is infinite.
Sixteen kilometres off the north tip of Western Australia, Troughton Island has a permanent human population of just two. It is less than one km2 in area, and although the island has an airport and an automated weather station, it has no standing drinkable water. Is it the sort of place where a population of Eurasian Tree Sparrows might persist?
Between 6 and 8 August, 2016, a survey of sea turtle nesting on Troughton Island was conducted by Uunguu Rangers from the Wunambal Gaambera Aboriginal Corporation and staff of the Western Australia Department of Parks and Wildlife. At that time the island’s resident caretakers, Brad and Heather Newman, pointed out four small sparrows foraging in the grass near the airstrip. These were later identified as Eurasian Tree Sparrows. Another expedition later that month found these four sparrows drinking from a dripping air conditioner. Three of the four sparrows were killed. The two carcasses that could be retrieved were tested for avian influenza, and found to be free of the disease. The fourth sparrow was not seen after September of 2016, and had presumably died.
An earlier caretaker on the island, Peter King, had reported as many as seventeen Eurasian Tree Sparrows on the island in 2011 following a storm. King noted that some of the birds had bred.
Reporting on these events, Anton Tucker of the WA Department of Parks and Wildlife and his colleagues speculated on the origin of the Eurasian Tree Sparrows on Troughton. The closest population of this sparrow to Troughton Island is four hundred kilometres away in Indonesia. This bird species is thought to periodically take up residence in new lands after hitching rides on large ships, and virtually all earlier records of this species in Western Australian have been associated with ports. However, this form of dispersal seems unlikely in the case of the sparrows on Troughton Island. The island cannot accommodate large cargo ships, and is visited infrequently by supply ships. Instead, the seventeen Eurasian Tree Sparrows first seen on Troughton Island in 2011 were likely blown there by a substantial storm.
According to the Australian Bureau of Meterology, Cyclone Carlos blew across the north and west coasts of Australia between 13 and 26 February, 2011. Very heavy rains caused widespread flooding and damage to roads and properties in and around Darwin, and forced the evacuation of the community of Nauiyu.
Beyond heavy rains, the cyclone may have also brought sparrows from the north to Troughton, if only for a time. The lack of available drinking water, combined with the presence of predatory Children’s pythons almost guaranteed that the plucky Eurasian Tree Sparrow would not survive on the island indefinitely.
Tucker, A. D. 2017. Invasive Eurasian Tree Sparrows Passer montanus on Trougton Island in the North Kimberly of Western Australia: a cyclone-induced colonization attempt? Australian Field Ornithology 34:67-70.
Photo credits: Eurasian Tree Sparrow Macau photograph – www.pinterest.com; stamp – www.bird-stamps.org
A Traveller’s Guide to Feathers, Article 90 -
Bearded Vultures are Rebounding
22 April 2018
Large animals face big challenges in the modern world. Their need for abundant resources, often spread thinly, may require undisturbed habitat over a vast area. These requirements can put large creatures in conflict with the activities of humans. It may be much easier to conserve populations of shrews than of whales; of geckos than of crocodiles.
Even though Bearded Vultures have a comparatively wide range across Europe, Africa and Asia, breeding populations are often quite small, and with a spotty distribution. This large vulture is not considered to be endangered, but the IUCN does consider it to be near-threated, and with a declining population.
In the late 19th century, these birds were exterminated in the Alps of Europe. Almost a century later, efforts to reintroduce Bearded Vulture to the Alps proved successful. These efforts began in 1986, and thirty-seven individuals were released between 1991 and 2008. The first successful breeding was seen in 1997, and the population began to grow.
It is crucial to document the whys and wherefores of reintroduction efforts, whether successful or not, because these can help direct future efforts of a similar kind. We need to build on our successes, and avoid making the same mistakes twice. To that end, David Jenny of the Swiss Ornithological Institute and his Swiss and Italian coworkers set out to record the demographics of the reintroduced population of Bearded Vultures in the Central Alps. The study area comprised 6000 km2 of mountains and valleys, 500 and 4000 metres above sea level. The team sought vulture territories, and document breeding between 1997 and 2015. Hundreds of volunteers contributed observations made on formal surveys.
By 2015, nine breeding pairs of Bearded Vultures were known from France, along with ten pairs in Italy, twelve pairs in Switzerland, and three pairs in Austria. These breeding pairs, nesting on cliffs, had a clumped distribution that seemed to radiate outward from the original release sites. Even though young vultures, born in the wild, may travel very long distances on exploratory trips, they tend to return to the region of their birth when they are old enough to breed. Many new pairs establish territories further and further from the original release sites, and so the increase in population number also means an increase in the species’ range in the Alps.
Although emigration from the study site to distant sites appears to be low, and the population may face inbreeding problems in the future, reintroduction of the Bearded Vulture in Alps must surely be considered a success worthy of celebration. So many vultures occupied the Alps by 2008 that authorities were able to stop introducing new birds from captivity. Jenny et al. wrote: “Overall, the nucleus in the Central Alps contributes substantially to the resettled and growing Alpine population of the Bearded Vulture.” Well done.
Jenny, D., M. Kéry, P. Trotti and E. Bassi. 2018. Philopatry in a reintroduced population of Bearded Vultures Gypaetus barbatus in the Alps. Journal of Ornithology 159:507-515.
Photocredits: Bearded Vulture photograph and stamp – www.pinterest.com
A Traveller’s Guide to Feathers, Article 89:
Urban Ornithology
08 April 2018
Last year, while on a trip to Faro in southern Portugal, Sarah and Michael Farn had the opportunity to eat breakfast on the rooftop of their hotel while watching a pair of White Storks tend to their chicks. The nest, roughly constructed of large sticks, was situated on the rooftop of a church. Sarah wrote to me to ask how often this species of stork nests in the heart of cities in Europe.
Well, Sarah, it seems that White Storks are perfectly pleased to nest in and around human settlements, and that the association between humans and storks may date back to the Neolithic period. In a recent publication, Grzegorz Kopij of the Wrocław University of Environmental Life Sciences revealed quite a lot about this interaction in Poland.
The city of Wrocław appears to have a lot going for it. The community has nearly 650,000 residents, and a history that dates back well over 1000 years. It is home to many fine universities with a combined population of more than 130,000 students. Wrocław lies in a valley where four smaller rivers join the large Odra River. From Kopij’s description, it seems that the city and surrounding countryside is a marvelous amalgam of grassland, forests and recreational area. Regrettably, the air is among the most polluted in all of Europe, but otherwise it sounds like a pretty neat place.
The storks certainly seem to think so. Two nests were located within a kilometre of the city centre. Surveys of nests in Wrocław demonstrated considerable variation among years, with just five nests in 1999, but nineteen breeding pairs in 2004. Kopij attributed some of this variation to the availability of foraging opportunities. When grass was mowed along the city’s river systems, the breeding population increased, but fell again when the grassland was left to grow wild. It seems that years with good rainfall resulted in more food for the chicks than in drier years. On average, White Stork nests in Wrocław raised 2.65 chicks, just a shade less productive than nests in rural settings.
Kopij found that Wrocław is the only Polish city with a population in excess of 100,000 residents to have a large population of nesting White Storks. He believes that modest management of the landscape around cities could have a positive influence on this species. Describing the bird as a charismatic species, Kopij suggested that White Storks: “may attract attention of naturalists, ecologists and conservationists in urban environments, (and) facilitate directly citizen engagement with nature, environmental education and awareness.”
If you are like Sarah, and would like me to write a column about a type of bird that really interests you, or would like to know more about a particular topic in bird biology, please be in touch with me at glen@glenchilton.com.
Kopij, G. 2017. Changes in the number of nesting pairs and breeding success of the White Stork Ciconia ciconia in a large city and a neighbouring rural area in South-West Poland. Ornis Hungarica 25:109-115.
Photo Credits: www.pinterest.com
A Traveller’s Guide to Feathers, Article 88:
Which One is Not a Real Study of Birds?
01 April 2018
In the spirit of the first day of April, I present a challenge. Below you will find descriptions of three studies. Your task is to deduce which two studies described are legitimate, and which one is made up. Are you ready?
Study Number One:
It is no surprise to anyone that many species of birds migrate long distances between their breeding grounds and spots where they spend the non-breeding season. Snails, on the other hand, are almost perfectly constructed to stay exactly where they are. Until…As part of a long-term study of migratory birds in Louisiana, researchers routinely captured, measured and banded songbirds in forested habitat near Johnson Bayou. On their northward spring migration, large numbers of birds use this spot after crossing the Gulf of Mexico. Each captured bird was inspected thoroughly. The assessment of an Indigo Bunting revealed a number of snails attached to the bird’s breast, buried beneath its feathers.
If you suspect that this description is the phony, then you are mistaken. In a paper published in The Wilson Journal of Ornithology in 2017, Theodore Zenzal, Emily Lain and Michael Sellers of the University of Southern Mississippi concluded that small, hitchhiking snails may be able to take advantage of songbirds to travel great distances, expanding their ranges and colonizing previously unoccupied spots. Transportation of this sort could have serious ecological consequences if exotic species arrive, establish themselves, and become invasive.
Study Number Two:
Males of some species of birds, but not all, have a penis. Some of these penises are pretty impressive; I will leave it to you to search for information on the Argentine Lake Duck. Within a single species of duck, there is considerable seasonal variation in penis length; they become longer in spring, and shorter after the breeding season. However, not all drakes are equally well endowed. What factors promote the genitalia of some males over others? Could it be that a surplus of males would cause each male to grow a longer penis?
Ruddy Ducks and Lesser Scaup were studied in captivity in Connecticut. Females were housed either with a single male, or with multiple males, establishing a competitive situation. When faced with competition for mating opportunities, drakes grew longer penises than did those with a hen all to themselves. The situation was more complex among Ruddy Duck drakes. Males in groups grew their penises more quickly, but maintained that length for a briefer portion of the breeding season.
If you picked this study as the sham, then you would be incorrect. Patricia Brennan of Mount Holyoke College and her coworkers published the results of their study of Lesser Scaup and Ruddy Ducks in 2017 in the journal The Auk. It is possible that a longer penis allows a drake to deposit his sperm deeper in a hen’s reproductive tract, which could be important when males are competing with each other. The authors described the genital size of male Ruddy Ducks as “extreme,” and their mating system as “promiscuous.”
Study Number Three:
That leaves only the third study, right? Consider calcium carbonate. This chemical compound is found in three states. One of these, calcite, makes up the bulk of bird eggshells. A different state of calcium carbonate, called vaterite, is rare in nature, probably because it is not a particularly stable compound. Among the places that it can be found is kidney stones and gall stones. It can also be found as a thin, chalky layer on the outside of eggs of some bird species. That layer doesn’t seem to prevent water loss across the egg’s shell. Could it be that vaterite helps prevent shell breakage when two eggs bump into each other?
“Vaterite” doesn’t even sound like a real word, and yet, once again, the study described was completely legitimate. Steven Portugal of Royal Holloway University of London and his colleagues studied vaterite on the shells of the Greater Ani, a large and beautiful cuckoo from Central and South America. In a 2017 article in the journal Ibis, the researchers explained that that when two Greater Ani eggs bump into each other, the dent that results is less than the thickness of the vaterite layer. These findings suggest that the role of vaterite, at least in Greater Anis, is to make eggshells less vulnerable to fracturing
Just because something seems unlikely, that doesn’t make it untrue. Keep that in mind this April Fool’s Day.
Photo credits: Indigo Bunting stamp, Ruddy Duck and Greater Ani – www.pinterest.com
A Traveller’s Guide to Feathers, Article 87
Birds That Provide Ecological Services
18 March 2018
In response to a question from reader Ellen Gasser, last week’s entry considered declining populations of the American Kestrel, Falco sparverius. Another reader of A Traveller’s Guide to Feathers, John Cunningham, wrote to me this week, asking if I could write another piece about recent advances in our understanding of American Kestrels. John is a recent convert to the world of birds, and that little bird of prey was one of the first new birds he learned. I discovered a 2017 publication by Megan Shave and Catherine Lindell of Michigan State University that seemed particularly relevant.
In our attempts to keep the ever-increasing human population well fed, more and more original intact operating ecosystems are being turned over to agricultural land. This sort of conversion generally results in a landscape that is less complex than the habitat it replaced. For birds, one the greatest difficulties is the destruction of nesting opportunities, such as nesting cavities in old trees.
And this is where American Kestrels comes in. The diet of these birds consists mainly of insects and small birds and mammals, many of which are pests in orchards. These kestrels nest in cavities in trees. If insufficient natural opportunities exist, perhaps they would be willing to utilize artificial nest boxes. However, it would be unreasonable to expect the owners of orchards to erect nest boxes without providing evidence that kestrels will use them.
Researchers Shave and Lindell surveyed American Kestrels in a fruit-growing region of northwest Michigan which lacks suitable natural nesting cavities. The researchers hypothesized that sites with nest boxes would be more likely to have the beneficial kestrel in residence. Were they correct?
In the study area, American Kestrels took up residence in 93% of newly-erected nest boxes, and these nests were successful in 91% of cases, producing an average of four fledglings per nest. Nest boxes that were not used were probably too close to other nesting pairs. Of the food provided to the chicks, 81% were arthropods, and 13% were small mammals. Good news!
The publication of Shave and Lindell had a great number of other details, including methodology, the specific results of surveys, and mathematical modelling of data, but all of that distills to a simple message. American Kestrels, at least in this fruit-producing region of Michigan were more likely to colonize and persist at sites to which nest boxes had been added. The authors went on to say that: “Increasing kestrel presence in and around orchards could enhance ecosystem services provided by kestrels.” Most earlier studies of birds of prey in agricultural landscape have focused on Barn Owls, and a broader understanding is bound to be valuable.
With luck, John, many orchard owners will respond to this study by providing nesting opportunities to American Kestrels.
Shave, M. E., and C. A. Lindell. 2017. Occupancy modeling reveals territory-level effects of nest boxes on the presence, colonization, and persistence of a declining raptor in a fruit-growing region. PLoS One 12 (10): e0185701.
Photo credits: Audubon illustration of American Kestrels - auduboneditions.com; American Kestrel in nesting box - http://www.birdwatching-bliss.com
A Traveller’s Guide to Feathers, Article 86:
Birds of Prey in Challenging Times
11 March 2018
I recently asked readers of A Traveller’s Guide to Feathers to write to me, suggesting topics or species about they would like to know a little more. Ellen Gasser wrote to ask about populations of American Kestrels, saying: “My feeling from personal observation is that they are far less abundant in farm/ranch areas of Alberta than 30 years ago.” Here is what I found, Ellen.
My first port of call was the website of the International Union for the Conservation of Nature, and their Red List of Threatened Species. According to the IUCN, the American Kestrel is of “least concern” with a population that is apparently stable across its very wide distribution. These birds have a vast range across North, Central and South America. That doesn’t sound too bad at all.
 Digging deeper, I found that things are not quite so rosy for the American Kestrel as the IUCN seems to feel. In a commentary piece in the latest issue of the Journal of Raptor Research, Christopher McClure of the Peregrine Fund and his colleagues pointed out that this bird appears to have been in decline for decades. The word “appears” is an important one in this case, because different sources of information provide different answers to questions about kestrel abundance. Christmas Birds Counts (CBC) do not always agree with the North American Breeding Bird Survey (BBS), which sometimes contradicts the results of counts of birds during migration. McClure et al. pointed out that conservation practices are only likely to be effective when we know the causes of decline, if such declines actually exist. What we need is evidence.
Digging deeper, I found that things are not quite so rosy for the American Kestrel as the IUCN seems to feel. In a commentary piece in the latest issue of the Journal of Raptor Research, Christopher McClure of the Peregrine Fund and his colleagues pointed out that this bird appears to have been in decline for decades. The word “appears” is an important one in this case, because different sources of information provide different answers to questions about kestrel abundance. Christmas Birds Counts (CBC) do not always agree with the North American Breeding Bird Survey (BBS), which sometimes contradicts the results of counts of birds during migration. McClure et al. pointed out that conservation practices are only likely to be effective when we know the causes of decline, if such declines actually exist. What we need is evidence.
Different methods of assessment of population abundance are almost certain to provide different answers. For instance, changes in numbers seen on CBCs may reflect changes in migration distance, rather than a change in the absolute number of birds. If surveys are conducted at the same time each year, changes in the seasonality of activity of birds brought about by climate change may be misinterpreted as a change in abundance.
If populations of American Kestrel are, in fact, falling, what is the most likely cause of the decline? Cooper’s Hawks, which prey upon the smaller American Kestrel, are increasing in numbers in some places. However, the researchers concluded that the timing of increases in the predators did not coincide well with declines in the prey. Could pesticides be causing kestrel declines? It seems that the use of the most-likely pesticides happened after kestrels began to decline in number. Might the alteration of kestrel breeding habitat be to blame, with fewer nesting cavities available to the birds? This seems unlikely because the numbers of American Kestrels have fallen even in spots where the occupation of artificial nest boxes, suitable for kestrels, has declined.
McClure and his colleagues suggested that several threats may be working together, interacting in some subtle way to produce the apparent decline in numbers of American Kestrels across North America. In trying to come to a concise statement about the state of our understanding, they wrote: “consistently too many kestrels seem to be dying relative to the number fledged each year.”
Rather than leaving their assessment at that rather unsatisfactory point, McClure et al. suggested a path forward. That path will not be an easy one, and results will not come quickly. It was suggested that the findings of count data, such as those of CBCs and the BBS should be supplemented by specific research on the abundance of American Kestrel’s across their range. Further investigation of the best ways to interpret nest-box studies are needed to avoid misinterpretation of results. BPheyond simple counts, the future of American Kestrels will rely on a better understanding of reproductive rates, dispersal of young individuals, adult survival, and specific causes of mortality. Information about the lives of these birds in the breeding season is a good start, but conservation efforts are most likely to be effective when we have also documented opportunities and threats across the kestrel’s range in the non-breeding season.
Thank you for the question, Ellen. American Kestrels do appear to be in decline, but researchers are working diligently to ensure their long-term survival.
McClure, C. J. W., S. E. Schulwitz, R. Van Buskirk, B. P. Pauli, and J. A. Heath. 2017. Commentary: Research recommendations for understanding the decline of American Kestrels (Falco sparverius) across much of North America. Journal of Raptor Research 51:455-464.
Photo credits: American Kestral stamp – www.canadianpostagestamps.ca; American Kestrel at a nesting hole – blog.kittykono.com
A Traveller’s Guide to Feathers, Article 85
Brave Birds
04 March 2018
Some birds are willing to take on tasks that would make a human stagger. Consider the American Golden-plover. Each year, these shorebirds migrate from their breeding grounds in Arctic Canada to spend the non-breeding season on the treeless plains of Argentina and Uruguay, and fly back again. Covering these huge distances, the American Golden-plover weighs about as much as a baseball or a cricket ball. That is an impressive feat.
These brave birds cannot cover those distances in a single long flight. They must stop along the way to rest, feed, and replace some of their feathers. On their northward migration, American Golden-Plovers may stop for two weeks or more to replenish themselves on the previous year’s leftovers in corn and soybean fields. It has been estimated that, at the peak of migration, a single county in a state south of the Great Lakes might host 25,000 individuals.
We all recognize the importance of renewable and non-depletable sources of energy. Parts of the globe seem ideally suited for wind-energy turbines, for instance. Even though the wind cannot be used up, those turbines might come with an environmental cost. Might those structures have an impact on migrating birds?
Wesley Homoya and his colleagues at Purdue University studied the responses of American Golden-plovers to wind energy facilities in west-central Indiana. More than four hundred turbines were constructed on their study site between 2008 and 2009, where none existed before. This sort of development seems likely to continue, and so an understanding of potential impact is crucial, particularly since the bird is a species of special concern in Indiana. Might turbines cause American Golden-plovers to avoid prime habitat? Might collisions with turbine blades kill birds?
Homoya and his crew surveyed agricultural fields in Benton County twenty-five times through the spring migration period. They estimated the number of individuals in each flock, and noted the time and location of each flock, as well as the type of crop from the previous season. The team spotted more than 100 flocks containing nearly 10,000 individuals. The distribution and abundance of American Golden-plovers was apparently only modestly influenced by the presence of turbines, and the research did not reveal strong avoidance on the geographic scales that they tested.
Some earlier studies found that wind-energy developments had negative consequences for migratory birds, but others were unable to demonstrate a significant impact. Until we have a better understanding of the full effects of wind-energy developments on bird populations, Homoya et al. suggest that we set aside those regions that have been identified as being particularly important to migrants. This might give many brave birds a better probability of survival.
Homoya, W., J. W. Moore, P. J. Ruhl and J. B. Dunning, Jr. 2017. Do American Golden-plovers (Pluvialis dominica) avoid wind-energy turbines in agricultural fields in Indiana during spring migration? Wilson Journal of Ornithology 129:863-871.
Photo credits: www.pinterest.com
A Traveller’s Guide to Feathers # 84
What Do You Most Want to Know About Birds?
25 February 2018
At no time in history has there been more scientists. Some claim that the number of active scientists exceeds the total number of scientists that worked in all previous generations combined. Researchers are making remarkable breakthroughs in all branches of the natural sciences. Some of these novel findings are the result of better technology, but most of the big discoveries are the result of the dedicated effort of lots and lots of clever, enthusiastic people. Hundreds of scholarly journals report on the results of those efforts. It is kind of sad, but the best that any scientist can do is try to avoid falling further and further behind.
This applies as much to birds as it does to quantum mechanics or planets circling distant stars. I try to keep up with the ornithological literature by reading at least the abstracts of every article in each of the journals that cross my desk. If the article is particularly relevant to my areas of study, or if I think it would make an interesting entry for A Traveller’s Guide to Feathers, I study the rest of the document.
To this point, I have been making guesses about what aspects of ornithological research might be most interesting to bird enthusiasts. The use of gopher tortoise burrows by birds on military sites? Perhaps. The responses of Golden Eagles to off-road vehicles? Maybe. I would like to move on from guesses, and create summaries of recent research concerning birds based on suggestions from readers.
And so, if there is a particular topic that you would like to know more about, please send me a suggestion. I will do my best to find results from a recent study, and let you know about them in a future installment of A Traveller’s Guide to Feathers. Are you keen to know more about the challenges of migration, or the ways in which birds respond to food shortages? Send me an email message. Is there a group of birds that particularly fascinates you, or even a particular species? Please be in touch.
Is there is a young bird enthusiast in your life, with a question about our feathered friends? “How come not all birds fly? Why is bird poop white? What is the smartest bird?” No matter how simple or complex the question, I will do my best to answer it.
Please write to me at glen@glenchilton.com, and I will try to address your interests in a future entry to A Traveller’s Guide to Feathers.
Photo credits: Migrating Cranes – www.pinterest.com; Golden Eagle by Daniel Hernanz – www.pinterest.com
A Traveller’s Guide to Feathers, Article 83 -
The Value of Introduced Populations
18 February 2018
Some types of birds have an enormous distribution, and so have the opportunity to bring joy to people in many countries. These include House Sparrows, Peregrine Falcons and White Wagtails, each of which can be found over large parts of the globe. At the other end of the spectrum are those species with a distribution so small that they barely exist at all.
Mariana Swiftlets are native to just the island of Guam and the Northern Mariana islands of Rota, Aguiguan, Tinian and Saipan. They are no longer found on Tinian or Rota. These birds nest colonially within caves, and forage for small insects over forests and grasslands. The species is endangered and with a decreasing population. According to the IUCN, these swiftlets face a number of challenges, including the use of pesticide which diminishes their prey, and exotic cockroaches which degrade their nests. An introduced mud dauber wasp also creates hardships for the swiftlet; by gluing their own weighty nests to the nest of the birds, the whole assembly may detach from the cave wall and crash to the ground. Introduced predatory brown tree snakes are now abundant on Guam, and are becoming established on Saipan.
Effecting conservation programs directed toward threatened species must be based on a thorough knowledge of the natural history of the imperiled animal. This leaves biologists in a bind. How do you study a threatened creature when the act of studying it may represent a damaging disturbance? Plans to reintroduce the Mariana Swiftlet to islands from which it has been eliminated are more likely to be successful if the bird’s biology is well known.
Nathan Johnson and his colleagues recently took advantage of a unique opportunity to study the Mariana Swiftlet without causing any harm to the bird in its native range. The species was introduced to the Hawaiian island of O’ahu in 1962 and 1965, and a small population still persists there. Between 2006 and 2011, the team studied swiftlets nesting in a human-created, 55 metre long tunnel that had been constructed before WWII. Using night-vision goggles and sensitive camcorders, the group monitored active nests in the tunnel. They also trapped rats inside and outside the tunnel.
In the O’ahu tunnel, active swiftlet nests were found in all months of the year. Each nest was home to a single egg, and hatching followed 23 days of incubation. The parents had a very good average nest success of 64%. Females were capable of more than one breeding attempt in a year. Rats were found to be a significant source of swiftlet mortality.
In order for the Mariana Swiftlet to be downlisted from endangered to threatened, authorities feel that there should be established populations of at least 1000 individuals on Aguiguan, and 2000 each on Guam, Rota and Saipan. It is hoped that studies of the swiftlet at its introduced home in Hawaii might better inform reintroduction practices. Birds from O’ahu might even be used to enhance populations on their native Mariana Islands.
Johnson, N. C., S. M. Haig, S. M. Mosher and J. P. Hellenbeck. 2017. Reproductive success of Mariana Swiftlets (Aerodramus bartschi) on the Hawaiian island of O’ahu. Journal of Field Ornithology 88:362-373.
Photo credits: Nesting Swiftlet – www.pinterest.com, originally posted on the Bishop Museum’s Hawaii Bird Survey website at hbs.bishopmuseum.org; artwork – www.pinterest.com
A Traveller’s Guide to Feathers, Article 82 – Loving Birds for Valentine’s Day. 11 February 2018
According to the Oxford English Dictionary, the word “valentine” was, at least in the mid-19th century, used as a verb. Valentine referred to the propensity of birds to greet one another by singing, particularly in the breeding season. The British poet George Meredith (1828 – 1909) used the word in a poem in 1851, writing: “Shall the birds in vain then valentine their sweethearts?”
The word “love” is used rather frequently in the naming of birds. There are, for instance, at least nine species of lovebirds, including the Black-cheeked Lovebird, Black-collared Lovebird, and Black-winged Lovebird. The genus name of these lovebirds, Agapornis, is based on Greek words meaning “love bird.” In celebration of Valentine’s Day I might have chosen to write about the Leaf-love, Phyllastrephus scandens, a reasonably common bird of central Africa. Regrettably, I was unable to find any recent studies concerning that species. The Brotherly-love Greenlet looked promising, until it became apparent that that was simply an archaic common name for the Philadelphia Vireo.
Then I came across a recent article by Gerald Mayr of the Natural History Museum in Frankfurt, and Paul Schofield of the Canterbury Museum in Christchurch. The paper described for the first time a New Zealand bird with no common name, but with the enchanting scientific name Australornis lovei. Please do not book a flight to New Zealand in hopes of adding a new bird to your life list, as A. lovei lived and died in the early Paleogene, some 61 million years ago.
The fossilized remains of A. lovei were collected from a locality near the Waipara River on the South Island of New Zealand. The bird’s genus name, Australornis, is descriptive, if not imaginative. It is derived from Latin and Greek words meaning “southern bird.” The species name, lovei, was used to honor the person who discovered the fossil in 2009, Leigh Love.
The fossilized remains discovered by Love constitute part of a wing, and the bones of the pectoral apparatus. All of the bones appear to be from a single individual. Most fossilized bird skeletons from 60-odd million years ago, and collected from the Australasian region, are clearly either a penguin or a penguin-like bird. Whatever else it might have been, A. lovei was not a penguin. Among the nine distinctive features of the skeleton are: “humerus with shaft cradiocaudally flattened,” “very long crista deltopectorlis,” and “ulna with caudal surface of proximal shaft forming a sharp ridge.” The specimen has features that are similar to those of a shorebird, an ibis, a duck, or perhaps a grebe. Even though Australornis is distinctive, it is not possible to narrow it down to a particular bird group. The remains are simply not sufficiently complete.
It seems unlikely that the wings of Australornis made it capable of gliding over long distances like an albatross, nor was it likely to have used its wings for underwater propulsion like a penguin. What did it do instead? With luck, future study will reveal additional skeletal components of this lovely bird, so that we will be able to infer more about its role in the world.
Mayr, G., and R. P. Scofield. 2014. First diagnosable non-sphenisciform bird from the early Paleocene of New Zealand. J. Roy. Soc. New Zealand. 44:48-56.
Photo credit: Black-cheeked Lovebird stamp; Lovebirds – www.pinterest.com
A Traveller’s Guide to Feathers, Article 81: If Not Monogamy, Why Not? 04 February 2018
Among animals, males and females in the vast, vast, vast majority of species do not form long-term pair bonds. Even when the field is narrowed down to animals with a backbone, the most common situation by far is for males and females to come together only long enough to mate. When there is an exception, an explanation is required.
Birds represent an exception. At least on the surface, birds of most species are monogamous, with males and females remaining together for an extended period, even in successive breeding seasons. The most frequently cited reason this prolonged pair bond is that both the father and mother are able to assist with raising the chicks to independence, even if the parents do not contribute in exactly the same way.
Biology is all about exceptions to exceptions, and our curious nature demands that these cases receive special consideration. If not monogamy, why not?
Juan Antonio Gil of the Fundación para la Conservación del Quebrantahuesos (Foundation for the Conservation of the Bearded Vulture) and his Spanish colleagues recently reported on a rare but interesting aspect of the natural history of Bearded Vultures. Also known as Lammergeiers, these birds most frequently breed as monogamous pairs. Polyandry, the situation in which a the breeding pair is joined by an additional male, is not at all uncommon amongst Bearded Vultures. The report of Gil et al. concerned the previously-unreported situation of polygyny in this species.
Studying Bearded Vultures in the mountainous southern slopes of the central Pyrenees, the research team observed three examples of “reproductive units” with more than one female. In the first case, a male and female were in a monogamous relationship from 2006 to 2012. In that interval, the female produced six clutches, but only two chicks hatched, and neither survived long enough to leave the nest. This pair were joined in 2013 by an additional female, but the trio produced no eggs. The first female departed in 2014, and the remaining pair successfully raised a chick in 2015. Through 2017, the first female remained in the area, but without a mate.
The second observed case of polygamy began with a pair that were monogamously mated from 2007 to 2009. They were not successful in that period. An additional female joined the pair in 2010, and an additional male did so in 2011. Through the 2017 breeding season, the foursome had no reproductive success.
The third case observed by Gil and his colleagues began when a female, five years old, occupied a breeding territory in 1999. She remained alone until 2004 when she was joined by a male. That pair built their first nest in 2007. An additional female joined the pair in 2011. The trio produced its first clutch of eggs inn 2016, but the breeding effort was a failure. The same three birds occupied the site in 2017, including the first female, now 22 years old. In all that time no chicks were fledged.
Why, then, are Bearded Vultures not all mated monogamously? An unbalanced sex ratio might explain the behaviour, but that was not the case in the Pyrenees population. Perhaps superior breeding habitat is limited, and so trios and even quartets form as an alternative to breeding on poorer territories. Again, that does not seem to be the correct explanation, because the polygynous groups did not occupy high-quality territories. The poor reproductive performance of the polygynous vultures suggests that the additional individuals were not gaining access to very high quality territories or very high quality mates.
Instead, Gil et al. felt that habitat saturation could be at play. Older birds without access to a breeding territory might be forced to join an already-mated pair “as an extreme strategy to produce offspring at any cost,” before they became too old to breed.
It is no wonder that Bearded Vultures need a conservation foundation.
Gil, J. A., G. Chéliz, Í. Zubergoitia and P. López-López. 2017. First cases of polygyny for the Bearded Vulture Gypaetus barbatus in the central Pyrenees. Bird Study 64:565-568.
Photo credits: www.pinterest.com
A Traveller’s Guide to Feathers, Article 80: A Plain Bird in Changing Times (28 January 2018)
To me, curlews, turnstones, sandpipers and snipe all look the same, varying only in their size. They all have long bills, reasonably long legs, and cryptic plumage. They wade in shallow water, and use their long bills to find food in the mud beneath their feet. Show me a bird enthusiast with a particular fondness for shorebirds, and I will show you someone who likes a challenge.
This isn’t a particularly charitable view, I suppose, but it seems reasonably astute. Take the Latham’s Snipe, for instance. The Field Guide to the Birds of Australia by Simpson and Day describes the body as cryptically mottled, the belly as pale, and the crown, eye-stripe and cheek stripe as dark brown. In the non-breeding season, Latham’s Snipes get even plainer.
This lack of pizzazz doesn’t mean that Latham’s Snipe doesn’t perform important ecological roles, nor that we can afford to ignore their long-term security. Luckily, the species isn’t currently threatened, with a global population estimated at between 25,000 and 100,000 individuals, but its numbers are in decline. According to the IUCN, the principal reasons for the bird’s decline are the destruction and degradation of its habitat. And what about global climate change? Are long-term changes in weather having an impact of the natural history of Latham’s Snipe?
David Wilson, Birgita Hansen, Jodie Honan and Richard Chamberlain, all residents of the state of Victoria in southeastern Australia, recently reported on their analysis of a wonderfully thorough data set. Latham’s Snipe breeds in Japan and far eastern Russia, but migrate to eastern Australia for the non-breeding season. Given that mean annual temperature and rainfall on the breeding grounds in Japan, and in the non-breeding areas in Australia, have changed significantly over the last century, Wilson and his colleagues asked whether Latham’s Snipe has altered the timing of its arrival in Australia.
The researchers used a range of sources of information, with particular reference to New South Wales (NSW) and the Australian Capital Territory (ACT). They scoured newspaper reports of the birds taken by hunters from 1846 to 1940. Thereafter, records of the first arrivals of Latham’s Snipe were taken from the Atlas of Living Australia, as well as newsletters, annual reports, and postings to online discussion groups devoted to birds.
The first reports of Latham’s Snipes in NSW and the ACT came as early as 30 July and as late as 23 September, with an average date of 14 August. Wilson et al. found no significant change in the arrival date over the 170 year study period. Although the first bird reported each year was not necessarily the very first individual to arrive, there is likely a strong correlation between the dates. In years gone by, a measure of honour befell the hunter who shot the first snipe of the season. More recently, dedicated bird enthusiasts have kept detailed records, and have been pleased to share their observations. The data are also likely to be skewed toward failed breeders who likely leave the breeding grounds before more successful individuals. Even so, the observations collected appear to represent a solid set.
Year-to-year variation in the timing of the first Latham’s Snipe in NSW and the ACT are likely to result from conditions on the breeding grounds and stopover areas. Even so, over the past 170 years, the long term average has not changed significantly. Are there comparably long-term data sets in your part of the world that might yield interesting findings about the timing of the lives of birds?
Wilson, D., B. Hansen, J. Honan and R. Chamberlain. 2017. 170 years of Latham’s Snipe Gallinago hardwickii arrivals in New South Wales and the Australian Capital Territory show no change in arrival date. Australian Field Ornithology 34: 76-79.
Photo credits: Latham’s Snipe drawing - http://animal.memozee.com/ArchOLD-6/1188176402.jpg & Wikipedia; Latham’s Snipe stamp - www.hipstamp.com
A Traveller’s Guide to Feathers, Article 79 – Showing Off to the Neighbours (21 January 2018)
From the Antillean Crested Hummingbird, through the Blond-crested Woodpecker, and on to the Crested Goshawk, there really do seem to be a lot of birds with feathery crests. In most cases, the bird can choose to lay its crest back, or erect it to best effect. It is fair to assume that birds use their crests as part of a display, but who is the intended recipient of the signal?
Spanish researchers Magdalena Ruiz-Rodríguez, Manuel Martín-Vivaldi, and Jesús Avilés of the Estación Experimental de Zonas Aridas and the Universidad de Granada investigated the use of crest displays by the Eurasian Hoopoe. Beautiful enough when resting quietly, these birds reveal starling black and white banding across the wings, back and tail in flight. When they get excited, both males and females show off their crest composed of long cinnamon/orange-coloured feathers with large black spots. Who are Eurasian Hoopoes showing off to?
Using male Eurasian Hoopoes, born in captivity, Ruiz-Rodríguez and her colleagues documented the displaying of crests when exposed to the playback of four sounds. The first sound was the singing of another male hoopoe, which might represent a rival. The second was a human voice, which could be perceived as a potential predator. As controls, the male hoopoes also heard the playback of background noise, and a blackbird, neither of which represent a threat.
The researchers found that the test subjects were significantly more likely to erect their crests when they heard playback of human voices (60% of trials), and in response to the playback of hoopoe songs (65% of trials), than they were to either of the control sounds (about 20% of trials).
It seems clear that the hoopoe display involving raising of crest feathers is directed at members of its own species, but also in response to potential predators. In terms of response to the playback of songs of its own species, males may perceive other males as rivals in the competition for mates. “The most probably target of the crest display in this situation,” wrote the researchers, “was the rival male.”
The behavioural response by the test hoopoes to the sounds of human voices has at least two possible interpretations. By erecting its crown, a hoopoe may be signaling to the potential predator, indicating that it has already been detected, and so the likelihood of a successful attack is small. Furthermore, hoopoes are thought to be unpalatable, presumably because of chemical secretions of their preen gland. Raptors are known to avoid Eurasian Hoopoes, and by erecting its crest a hoopoe may be making its identity very clear. Alternatively, the display utilizing an erect crown may not be directed at the potential predator per se, but rather at other nearby hoopoes. The display may be a way of saying: “Have you noticed that predator yet?”
In the animal kingdom, signals tend to be very specific. There are not many examples of individuals using a single display in two entirely different contexts. The dual-functionality of the crest display of Eurasian Hoopoes may lead ornithologists to look for more cases.
Ruiz-Rodríguez, M., M. Martín-Vivaldi and J. M. Avilés. 2017. Multi-function crest display in hoopoes Upupa epops. Journal of Avian Biology 48:1425-1431.
Photo credits: www.pinterest.com
A Traveller’s Guide to Feathers, Article 78 – Enjoying a Cup of Coffee While Helping Birds (14 January 2018)
Each of us has endless opportunity to make small decisions with positive results. The ethical decisions of a single individual are unlikely to make a large difference, but when we start acting collectively the world becomes a better place. Many of those positive decisions are related to our purchase of everyday products, and your choice of coffee may help our feathered friends.
Traditionally, coffee plants were grown under shade trees. This situation changed when varieties of coffee were developed that had better productivity when exposed to full sunlight; most coffee is now grown without shade trees. Over the past couple of decades, research has shown that coffee plantations with abundant shade provide habitat that is far more valuable to insect-eating birds. These include several species of New World warblers that breed in temperate regions but spend the non-breeding seasons in the tropics. This relationship is not all one-way. Warblers eat insects, and it is apparent that their foraging benefits the coffee farmer by eliminating pests. With support from such agencies as the Smithsonian Migratory Bird Centre, the popularity of shade-gown, bird-friendly coffee is growing.
Aaron Spidal and Matthew Johnson of Humboldt State University in Arcata, California recently asked a very interesting question about migrant warblers and coffee farms. Do all members of a particular species of warbler have equal access to the high-quality foraging opportunities provided by shaded coffee farms, or are some individuals forced to live on sunny farms? Might older birds, or behaviourally-dominant males, prevent females and young males from feeding in shady situations?
In December and January of 2010-2011 and 2011-2012, Spidal and Johnson mist-netted birds at coffee farms in Jamaica. The nine sites chosen for study differed greatly in the amount of cover, from highly shaded to fully sunlit. Nearly 1400 hours of trapping resulted in 298 captures of migratory warblers of 14 species. The most abundant of these were the Black-throated Blue Warbler, the American Redstart and the Prairie Warbler.
The researchers found that coffee farms with differing amounts of shade did not differ in the number of adult and young birds, by any particular species or for all species combined. However, there was a significant positive relationship between the proportion of males at a site and its cover by shade trees. This was true for each of the three most common warbler species, and for all species combined. Shady conditions provided better foraging opportunities, and males occupied those sites more frequently than females.
The sexual subdivision of habitat seen by Spidal and Johnson might be a result of sexual differences in habitat preference. Perhaps there is something special about the anatomy or behaviour of females that make foraging in sunny coffee farms the better choice. The researchers favored the alternative explanation that males were dominant, and so excluded females from the superior foraging opportunities provided by shady coffee farms. Although further study would be required, Spidal and Johnson felt that the proportion of males at a Jamaican coffee farm might serve as an index of habitat quality.
At present, it is unlikely that you will find shade-grown, bird-friendly coffee in your local grocery store. If bird-loving consumers make their wishes known, that situation is likely to change in the years to come.
Spidal, A. B., and M. D. Johnson. 2016. Sexual habitat segregation in migrant warblers along a shade gradient of Jamaican coffee farms. Journal of Caribbean Ornithology 29:37-42.
Photo credits: Black-throated Blue Warbler – www.pinterest.com; Shade-grown coffee plantation - www.alamy.com/stock-photo/coffee-plantation-landscape.html
A Traveller’s Guide to Feathers, Article 77 – Observations of an Elusive Bird (07 January 2018)
When it comes to wildlife, ignorance is never bliss. Decisions about conservation issues are only likely to be effective if they are based on a thorough understanding of the natural history of threatened species, and of the reasons for declines in their numbers.
Indonesia has more threatened species of birds than any country other than Brazil. As in many other parts of the world, Indonesian birds are being lost as a consequence of deforestation resulting from increasing demand for land for agriculture. There is, however, an additional threat to the birds of Indonesia. The nation has a long history of keeping birds as pets, and this causes a very high demand for birds taken from the wild. Although laws exist to protect species like the Sumatran Laughingthrush from exploitation for the pet trade, enforcement of those laws is sporadic in Indonesia, and birds are sold openly in markets by vendors who are unlikely to be prosecuted.
It is against this bleak backdrop that Tomáš Bušina and Marek Kouba added their observations of the behaviour of the Sumatran Laughingthrush. An endemic to the island of Sumatra, this laughingthrush is considered by the International Union for the Conservation of Nature to be endangered and with a declining population. Formerly widespread, the bird can now only be found in remote mountain refuges. The inaccessibility of these spots has hampered study of the Sumatran Laughingthrush, and fundamental aspects of the bird’s natural history remain unknown.
The study site of Bušina and Kouba was in the Karo regency of North Sumatra, at an elevation of 1600 metres. The authors withheld the precise coordinates of the site for obvious reasons. They described the locale as a “rugged montane landscape shaped by valleys with small rivulets… densely covered by trees, with a closed canopy height up to 35 m.” Ideally Bušina and Kouba would have captured laughingthrushes in mistnets, and marked them for individual identification before fitting them with small transmitters for tracking. Unfortunately, the birds could not be caught in nets, and so the researchers devised a different study protocol.
Three observation hides were constructed, and birds were then spotted for twelve hours on each of 21 days between late 2014 and early 2015. Amoung the observations recorded were number of individuals, their behaviour and their group structure.
Groups of Sumatran Laughingthrushes were recorded seventy times. These groups never exceeded five individuals as they moved through the shrubbery, maintaining contact with each other using quiet vocalizations. Individuals were never spotted on the ground. At times, these threatened birds were spied in mixed-species feeding flocks along with Black and Chestnut-capped laughingthrushes, as well as Sumatran Drongos and Sumatran Trogons. These sorts of mixed-species gatherings are not uncommon among birds in the tropics. Although the researchers could not be certain, it seemed that all observations of Sumatran Laughingthrushes were of a single group.
I wrote to Bušina to ask how he came to study the Sumatran Laughingthrush in Indonesia, so far from his home in Europe. He said: “Everything started in 2012 when I completed my Master’s degree program and came to Indonesia as a volunteer. After one year of cooperation with a local NGO I became a project manager of its Sumatran Laughingthrush conservation program. At the same time I started my PhD focused on its restoration and black market monitoring.”
There are both joys and challenges to studying birds in remote spots. Bušina told me that the feeling of being swallowed up by the Sumatran montane rainforest is almost indescribable. “This deep dense jungle can cause a claustrophobic feeling, how silent and lifeless it can look, and then someone could be surprised by a huge number of foraging birds flying over the basecamp. Simply flabbergasting and overwhelming.” Despite the ferocious beauty, in the rainy season Bušina would have to remove as many as 100 leeches from his body each day.
When I asked him what he most wanted to know about the Sumatran Laughingthrush, Bušina indicated that it was hard to know where to begin a response. Although we can venture some guesses, most of the biology of this bird is completely unknown. “The most important thing to know is the current size of remaining population and its real distribution, because this species is under heavy trapping pressure resulting in considerable population decline. The Sumatran Laughingthrush is likely reaching the brink of extinction.”
Although the conclusions that can be drawn from this study of Sumatran Laughingthrushes are modest, the systematic observations of the elusive and endangered bird are the only ones available. Let’s hope that the work of Bušina and Kouba will inspire further study.
Bušina, T., and M. Kouba. 2017. Preliminary observations of the home range size and behaviour of the Sumatran Laughingthrush Garrulax bicolor. Kukila 20:30-38.
Photocredits: pair of Sumatran Laughingthrushes – www.cikanangawildlifecenter.com/wp-content/uploads/2013/06/Sumatran-Laughing-Thrushes-Garrulax-bicolor.jpg; Sumatran Trogon – www.pinterest.com
A Traveller’s Guide to Feathers, Article 76 – Has Anyone Got a Light? (31 December 2017)
As one year turns to the next, many of us will be resolving to make changes in our lives. A commitment to stop smoking in the new year is a time-honored tradition. However, that tradition may not be quite so well established in birds.
The lives of birds that live in cities attract the attention of many biologists. It seems that birds with a degree of behavioural flexibility, including the ability to exploit new resources and deal with novel challenges, are the species best able to take advantage of urban environments. Studies have shown that urban-dwelling birds consume novel foods, change the structure and pitch of their songs, and alter the timing of nesting, all to make the best of life in the city. Even so, some of the changes in bird behaviour seem a little peculiar.
Monserrat Suárez-Rodríguez and Constantino Macías Garcia of the Universidad Nacional Autónoma de México studied the use of cigarette butts by House Finches in the construction of their nests in Mexico City. Cigarette butts? Really? When this sort of behaviour has been seen in the past, it has been assumed that noxious chemicals remaining in the butt after the rest of the cigarette has been smoked may serve to reduce the infestation of ectoparasitic ticks, mites, fleas and so on. Intuitively, this explanation makes a lot of sense. We have long recognized the ability of ectoparasites to cause impaired incubation and brooding by parent birds and blood loss in their chicks. Furthermore, ectoparasites have the ability to transmit a wide range of nasty diseases from one bird to another.
Intuitive sense is one thing, but experimental evidence is another. After all, perhaps the cellulose fibres in a cigarette butt are utilized in nest construction because of their insulative value, and not for their ability to repel parasites. Suárez-Rodríguez and Garcia resolved to untangle the riddle.
Having identified a House Finch nest, the researchers waited for the resident female to finish laying her eggs. They then removed the lining of the nest, and replaced it with an artificial lining which was free of both ectoparasites and cigarette butts. Some of nests then received seventy live ticks, and some received seventy dead ticks. No ticks were added to a third group of nests.
Later, each nest was checked for the addition of cigarette butts by the parent birds. The results were clear. Those nests to which seventy live ticks were added subsequently received significantly more cigarette butts than nests which had dead ticks or were tick-free. Most of the butts had been woven into the lining of the nest. It seems that ectoparasites cause House Finches to add fibres from discarded cigarette butts to their nests, at least in Mexico City. Suárez-Rodríguez and Garcia described the behaviour of the finches as “self-medication.” Wild tobacco plants grow in Mexico City, but finches have not been seen to add leaves from these plants to their nests. It may be that butts are a better anti-parasite treatment than tobacco leaves themselves.
In the coming days, if you fail to keep to the resolutions for change that you have made, perhaps you can say that you are continuing on your destructive path for the sake of the birds.
Suárez-Rodríguez, M., and C. M. Garcia. 2017. An experimental demonstration that House Finches add cigarette butts in response to ectoparasites. Journal of Avian Biology 1316-1321.
Photo credits: House Finch hen collecting a cigarette butt, © Suárez-Rodriguez - www.sciencenewsforstudents.org; male House Finch © Judy Gallagher - http://www.flickr.com/photos/52450054@N04/6920300924/ (reproduced without modification by Creative Commons licence 2.0; https://creativecommons.org/licenses/by/2.0/)
A Traveller’s Guide to Feathers, Article 75 – Christmas Birds (24 December 2017)
It seems an appropriate time of year to spare a thought for William Mynors. An English seaman, it isn’t certain quite where or when he was born, nor where or when he died. Among the few things we do know about Mynors is that he captained the ship Royal Mary, and that on Christmas Day, 1643, he and his crew sailed past an island in the Indian Ocean without stopping. He named the feature Christmas Island, and more than three hundred and seventy years later, that is the name that we still use.
With a human population of about 2,000, Christmas Island is governed by Australia, although it is much closer to parts of Indonesia. The island has an area of about 130 square kilometres, much of which is designated as a protected area. And what of the birdlife on Christmas Island?
Avibase is a marvelous website, maintained by Dr. Denis Lepage of Bird Studies Canada. Whether I need to check the current thinking about the taxonomic status of a particular bird, or the number of bird species in a region, Avibase is generally my first stop. Avibase’s checklist of birds of Christmas Island provides some interesting statistics. For instance, 164 birds species are known from the island, but at least 115 of those are considered to be rare or accidental.
That leaves just forty-nine species that you might feel confident about seeing on Christmas Island. Four of those have been introduced to the island. The Java Sparrow, House Sparrow, Eurasian Tree Sparrow and Red Junglefowl all have a comparatively large global distribution because of introduction efforts in generations past. Of the remaining forty-five species, six strike me as being particularly interesting. If your life’s ambition is to see every type of bird in the world, it is these six which will require you to visit Christmas Island.
The island is home to two breeding endemics. These are seabirds that breed only on Christmas Island, although their wanderings in the non-breeding season take them elsewhere. The first is Abbott’s Booby which is considered endangered. The second is the Christmas Island Frigatebird, a critically-endangered species. The list of factors working against the frigatebird is a long one. On Christmas Island, their colonial nesting sites are damaged by dust from phosphate mining, and are vulnerable to cyclones. Introduced yellow crazy ants may become a problem for nesting birds in the future. In the non-breeding season, Christmas Island Frigatebirds are harmed by oceanic pollution and garbage, and they are hunted in Indonesia and Malaysia.
The Christmas Island Imperial-pigeon and the Christmas Island White-eye are both found on this particular island and nowhere else. Both species are considered to be near-threatened; even though the range of both are limited, they are comparatively abundant on the island. The Christmas Island Boobook, a lovely large owl, also an endemic, has a global population of between 500 and 1,000 mature individuals, and is considered vulnerable. The taxonomy of the Christmas Island Swiftlet is not certain. Whether a full species, found only on Christmas Island, or merely a subspecies of the Glossy Swiftlet with a much wider distribution, this birds seems to be performing reasonably well.
Wherever you are in the world, Lisa and I wish you a very Happy Christmas. We hope that your life is full of birds.
Photo credits: Abbott’s Booby stamp - www.birdtheme.org; Christmas Island White-eye stamp - www.koalastamps.com
A Traveller’s Guide to Feathers, Article 74 – Go Ahead, Impress Me (17 December 2017)
One of the most wonderful things about the study of bird biology is the endless means by which birds of different species deal with the challenges of survival and reproduction. It will be immediately apparent to all that males of many bird species are brightly coloured, and have elaborate ornaments that presumably serve to attract females. Male birds also display a wide range of ritualized behaviours that allow a female to choose the best male from among potential suitors. What is less apparent is that female birds, seeking out the best males, also go to great lengths to impress.
In a recent publication, Juan Rubalcaba of the Universidad Rey Juan Carlos and his colleagues reported on their study of the behaviours of a bird species with complex nesting behaviour. The Spotless Starling has a large breeding distribution, ranging through Portugal, Spain and Italy, and across northwestern Africa. Its scientific name, Sturnus unicolor, is based on a couple of Latin words that translate to English as “the starling that is kind of boring to look at.” It does however, express some interesting behaviours at breeding time.
Rubalcaba and his team described Spotless Starlings as: “facultatively colonial and moderately polygynous.” Both males and females attempt to defend the territory against intrusion by members of their own sex. More is not better in this case. Males perform defense behaviours at the start of the breeding season, and collect leaves, flowers and lichen to the decorate the nest, attracting and stimulating potential mates. Later in the season, during egg-laying and incubation, females utilize feathers from other bird species to decorate the nest, presumably signaling her quality to her mate, and perhaps to dissuade other females from moving in.
Spotless Starlings utilize both natural cavities and artificial nest boxes. This gave the researchers the opportunity to establish high-density and low-density breeding areas within the study area. Do males occupying territories in high-density breeding areas bring more vegetation to the nest? Do females in high-density breeding areas bring more feathers to the nest?
In their study area near the community of Manzanares el Real in Central Spain, the researchers found that the answer to both questions was yes. More birds means more vegetation and more feathers. Furthermore, although the average number of eggs produced in the two areas was the same, the probability of nestling surviving long enough to leave the nest was lower in the high-density area. Greater competition resulted in lower reproductive success.
It seems that the use of plants and feathers as nest decorations by male and female Spotless Starlings signals of their territorial nature. The researcher explained that “The amount of green nesting material and feathers are related to the condition of males and females, respectively, and they may reflect their competitive abilities or willingness to defend their territories against conspecific competitors.”
It isn’t just males that need to impress. It seems that females can also benefit from the use of decorations to signal their status and health to other birds in the vicinity.
Rubalcaba, J. G., Daniel Fuentes, J. P. Veiga and V. Polo. 2017. Nest decoration as social signals by males and females: greenery and feathers in starling colonies. Behavioral Ecology 28:1369-1375.
Photo credits: www.flikr.com & www.pinterest.com
A Traveller’s Guide to Feathers, Article 73 – Heavy Metal Birds (11 December 2017)
The Kagu is surely one of the most wonderful birds that you may not have had the opportunity to see yet. Looking a bit like a cross between a pigeon and a court-jester, Kagus are not particularly closely related to any other species of bird. They are found only in the rainforests of New Caledonia, an island-nation far off the east coast of Australia. The Kagu is an essentially flightless bird, and so they spend their days running through the undergrowth in search of earthworms which make up the bulk of their diet.
The climate of New Caledonia is not particularly seasonal, and so birds can breed throughout the year. When Kagus get around to breeding, the reproductive pair are assisted by one or more helpers. Despite this help, only one egg is produced in each breeding attempt. Kagu chicks leave the nest just three days following hatching, after which they receive two months of care from the attending adults. The good news is that Kagus have a stable population. The bad news is that they are listed as endangered, a result of a small global population and a limited range.
Jörn Theuerkauf of the Polish Academy of Sciences and his colleagues from institutes in Japan, Germany, the United States, and New Caledonia recently published the results of a study that investigated a peculiar aspect of the Kagu’s biology. It seems that at least one-third of New Caledonia has ultramafic soils. I dashed to the dictionary so that you don’t have to. Ultramafic soils are the results of weathering of igneous rock, and have particularly high concentrations of magnesium and iron. It doesn’t stop there; ultramafic soils can be expected to have high concentrations of other heavy metals such as vanadium, chromium, copper, nickel, mercury, and selenium. Many of these elements are essential to the health of Kagus, as they are to the health of humans, but only in trace quantities. If ingested quantities become too high, impaired health can be expected.
Between 2003 and 2012, Theuerkauf and his colleagues followed the breeding biology of twelve Kagu families in Parc Provincial de la Rivière Bleue where the soils are ultramafic, and seven families in Parc Provincial dess Grande Fougères where the soils do not contain high levels of heavy metals. Individual birds were tracked using radio transmitters and activity sensors. The researchers sought out nests, and videotaped breeding activities. Heavy metals can be sequestered in feathers, and so the team collected feathers from birds and analyzed the concentrations of twenty-seven different elements, from arsenic to zinc.
Not surprisingly, feathers collected from Kagus in the region with ultramafic soils had greater quantities of some heavy metals than feathers from birds in the spot with non-ultramafic soils. The former feathers had, for instance, thirty-one times more chromium, and nine times more cobalt than the latter feathers.
Furthermore, the reproductive output of Kagus in the provincial park without ultramafic soils was four times higher than those birds living on land with soil high in heavy metals. This latter result was almost entirely the result of the former birds laying more eggs in each season. Other aspects of the breeding biology, such as the mass of the eggs, and the proportion of chicks that survived, did not differ between the two spots.
That left Theuerkauf et al. to make a link between cause and effect. Why did Kagus on the heavy-metal rich soil produce fewer eggs? Given that Kagus are endangered, it would have been unethical to expose individuals to additional heavy metals, and observe the result. Were Kagus in Parc Provincial de la Rivière Bleue being poisoned by metals taken up by the earthworms that the bird feed upon? It seems not. Instead, high concentrations of heavy metals in the spot with ultramafic soils results in reduced soil fertility. This results in fewer invertebrates in the soil on which the Kagus feed. With lesser availability of food, it takes birds longer to replenish their reserves after each breeding attempt.
Work on threatened species almost always has conservation implications. In the case of Kagus, the results of the study by Theuerkauf and his team suggest that current efforts should probably be directed toward populations living in areas where the soil is not high in heavy metals. In those spots, birds are likely to grow in number more quickly.
It is through the efforts of researchers like Theuerkauf et al. that you may, one day, have the opportunity to see Kagus in the wild.
Theuerkauf, J. et al. 2017. Elevated concentrations of naturally-occurring heavy metals inversely correlate with reproductive output and body mass of the Kagu Rhynochetos jubatus. Ibis 159:580-587.
Photo credits: www.pinterest.com
A Traveller’s Guide to Feathers, Article 72 – Eat What?!? (03 December 2017)
Dunnocks are nondescript garden songbirds native to much of Europe and the Middle east, and can be found in parts of Asia. They have also been introduced to New Zealand, and it was there that Simon Lamb and his colleagues chose to study a peculiar aspect of avian biology.
For almost all species of birds, nest sanitation is crucial. Nestlings poop, and if the nest isn’t to become fouled and stinky, a situation that has the potential to attract predators, something must be done with all of that faeces. In order to make things easier for the parents, the nestlings of some species deposit their wastes in a sac which the adults can carry away from the nest.
Lamb et al. studied an odd behaviour associated with fecal sacs. Although it has not been studied very well, it seems that a lot of parent perching birds sometimes eat the faecal sacs of their chicks, rather than simply discarding them. The ingestion of waste products is known as coprophagy, and it is so remarkable that it requires an explanation.
As Lamb and his colleagues pointed out, two explanatory ideas have been proposed for coprophagy. The first of these suggests that by eating their chicks’ wastes, parents may be taking advantage of nutrients that the chicks did not absorb. This might be most relevant early in the nestling’s life, when its digestion system is not particularly efficient such that some nutrients pass right through. The alternative explanation suggests that the time and energy demands on parents with young chicks is almost certainly very high, and that swallowing faeces is a time-effective way of dealing with nest sanitation. It is possible, of course, that coprophagy is performed for both of these reasons.
Some Dunnocks choose to lead complicated lives. Monogamous partnerships are frequently observed, but so too are polyandrous triads in which a female is mated to two males. The direct costs of reproduction are higher for females than for males because the former build the nest, incubate the eggs and brood the chicks. Among their breeding-season tasks, males contribute to feeding the chicks. The New Zealand researchers made a number of predictions about the frequency of coprophagy by Dunnocks based on their reproductive behaviour.
The study was conducted on the botanic gardens inn Dunedin. Dunnocks were colour-banded for individual identification, and the comings and goings of birds at twenty-six nests were documented with video cameras.
Coprophagy was observed at nineteen of the twenty-six nests under scrutiny. Females were more likely to eat the faecal sacs of their chicks than were males. There were no statistical differences between the consumption of faecal sacs by either males or females in monogamous versus polyandrous partnerships. As the nestlings got older, coprophagy was observed less frequently. Although their results did not allow for a definitive answer about the reason why parent birds eat the faeces of their chicks, Lamb et al. felt that the parental nutritional hypothesis was best supported.
It is not yet clear what cues parents use in their decision to eat or discard a particular faecal sac. Smell… colour… texture… taste? That question will have to await future research. In the meantime, waste not, want not.
Lamb, S. D., et al. 2017. Coprophagy in Dunnocks (Prunella modularis): a frequent behavior in females, infrequent in males, and very unusual in nestlings. Wilson Journal of Ornithology 129:615-620.
Photo credit: Dunnet - upload.wikimedia.org
A Traveller’s Guide to Feathers, Article 71 – One Puffleg, More or Less (26 November 2017)
If you would like to make your day a little bit happier, use an online search engine to find images of the Black-breasted Puffleg. Knowing that this gorgeous little bird, one of 120 species of hummingbird in Ecuador, shares the world with us is almost certain to help make you glad to be alive.
If, instead, you are determined to be miserable today, then have a look at the IUCN’s description of the current state of the world for the Black-breasted Puffleg. You will read that “around 93% of the suitable habitat within its probable historic range has been degraded or destroyed… Human-induced fires threaten large tracts of forest during the dry season…The construction of a pipeline… led to habitat destruction for the pipeline itself, an access road and a depressurisation station despite the known presence of the hummingbird.”
With a population of less than 1000 individuals, and a global distribution of just sixty-eight square kilometres, all within Ecuador, the Black-breasted Puffleg is considered to be critically-endangered. Restoration of suitable habitat is seen as crucial to its ongoing survival. If the puffleg is to take advantage of that habitat that we might try to restore, then it is essential that we know its preferred foods, and whether it might be willing to use habitats that are in the process of being rejuvenated. Until recently, that knowledge was lacking.
Estaban Guevara of Aves y Conservación – BirdLife in Ecuador and his colleagues sought to redress that lack of information. They wanted to know which of the available flowering plants were exploited by the Black-breasted Puffleg for food, and determine patterns of use of habitats that differed in their level of disturbance.
The study was conducted at five sites of evergreen forest on the flanks of the Pichincha volcano in northwestern Ecuador. The researchers sought out flowers with characteristics that might make them suitable for foraging by the pufflegs, and used time-lapse cameras to determine which plants were visited most frequently.
One photographic image was captured every second over nearly 3000 hours of observations. These revealed 144 visits by Black-breasted Pufflegs to seven of the twenty-one species of flowering plants monitored. A plant known by the scientific name Macleania rupestris was the most frequently visited species at one site, and Palicourea fuchsioides at another site. As if things weren’t bad enough for the puffleg, Guevara et al. described P. fuchsioides as “an endangered shrub known only from a few locations in the Andes of western Ecuador.”
Black-breasted Pufflegs were observed at only two of the sites that appeared to the researchers to be most suitable for occupation, and the birds seemed to avoid highly disturbed habitats such as forest edges. The recovery of degraded habitat, and the reconnection of fragmented patches of good habitat may be possible, but a better understanding of the natural history of key flowing plant species will be needed.
It doesn’t matter to me that I will probably never get to see a Black-breasted Puffleg. It is enough for me to know that they exist. What is important to me is that people like Esteban Guevara and his colleagues care enough about the world to try to keep our birds from slipping away.
Guevara, E., R. Hipo. C. Poveda, B. Rojas, C. H. Graham, and T. G. Santander. 2017. Plant and habitat use by Black-breasted Pufflegs (Eriocnemis nigrivestis), a critically endangered hummingbird. Journal of Field Ornithology 88:229-235.
Photo credits: Black-breasted Puffleg, photograph © Murray Cooper – www.birdlife.org; Black-breasted Puffleg, photograph © Steve Blain – www.neotropicalbirds.cornell.edu
© Steve Blain
Week 70 – 19 November 2017
First, Last, or Somewhere in the Middle
Last week I described why it is to the advantage of parent birds to have all of their eggs hatch on the same day, even though their eggs are laid on different days. By waiting until the last egg is laid to begin incubation, all chicks in the nest will hatch synchronously, and will be at roughly the same stage of development. This is the most common situation among birds. But there are exceptions, and exceptions demand explanations.
Zebra Finches have a vast distribution in Australia, and are also found on the islands of Lombok, Timor and Sumba. As seedeaters they are fond of open habitat, and can breed at anytime of the year if water is available. They breed readily in captivity, and are a favourite of novice cage-bird fanciers. Curiously, Zebra Finches do not wait to begin incubation until after the last egg is laid, and so chicks in the nest are different ages. This is true of birds both in the wild and in captivity.
Maki Ikebuchi and colleagues at the University of Tokyo in Japan and Universität Bielefeld in Germany studied the phenomenon of asynchronous hatching of Zebra Finch chicks, searching for an interpretation of this peculiar behaviour. They utilized captive subjects, providing them with unlimited access to food and water, and appropriate materials for the construction of nests. In most cases, birds began breeding almost immediately.
Eggs hatched after 12 days of incubation, and Ikebuchi’s team documented the development and masses of each chick each day. Eighteen clutches of eggs resulted in sixty-three chicks. The average period over which all eggs in the clutch hatched was 3.1 days. Compared to some songbirds, development of the chicks required an extended residency in the nest. Their eyes did not begin to open until eight days after hatching, and the first proper feathers did not appear until day ten. Young birds began leaving the nest after eighteen days, and the oldest chicks returned to the nest over a period of days until their younger siblings were also ready to fledge.
In these captive Zebra Finces, asynchronous hatching resulted in an unlevel playing field, with the first-hatched chicks gaining weight faster, and ultimately weighing more than chicks that hatched later. The difference was, curiously not very large. Furthermore, the researchers found that the probability of survival of chicks was not influenced by their place in the hatching order. The researchers considered the possibility that, through differential provisioning, parents might actively seek to maintain the differences in weight among their chicks. If so, the parents seem to have been defeated in their goal. Given that the ultimate differences in weight were quite small, Ikebuchi et al. speculated that the parents may be unable to discriminate between older and younger chicks at feeding time, perhaps because the interior of the nests is quite dim.
In birds such as eagles and pelicans, eggs do not hatch synchronously, and chicks from the first-laid eggs are more likely to thrive than chicks from the last-laid eggs. Even though Zebra Finches also show asynchronous hatching, not commonly seen among songbirds, there appears to be no particular advantage to hatching first, at least in captivity. It will be interesting to see if future research on wild populations of Zebra Finches has the same result.
Ikebuchi, M., K. Okanoya, T. Hasegawa, and H.-S. Bischof. 2017. Chick development and asynchronous hatching in the Zebra Finch (Taeniopygia guttata castanotis). Zoological Science 34:369-376.
Photo credits: ‘wild-type’ captive Zebra Finches on a nest – www.singing-wings-aviary.com; one of the many forms of exotic colouration in Zebra Finches in captivity - https://www.youtube.com/watch?v=SUaeeyNMCUI .
Week 69 – 12 November 2017:
Except for Eagles and Pelicans…
One of the themes of this series is that we generally know more about the lives of birds than we do about any other group of animals. Most birds are active during the day, and are not shy about their comings and goings. Many incubate their eggs and feed their chicks in places that are easy to access, and some breed readily in captivity. Some birds are particularly good indicators of environmental health. If you wish to investigate underlying truths about life, birds make ideal study organisms.
Among the phenomena that have been particularly well studied in birds is the timing of the start of continuous incubation of the eggs. Even the best fed hen can only produce one egg a day. As a result, eggs in a nest are bound to be different ages. If the parent birds were to begin incubation as soon as the first egg was laid, embryonic development of that first egg would begin right away, and chicks would hatch on different days, resulting in chicks of different sizes.
There are some spectacular examples of how chicks of different ages works perfectly for the parents, ensuring that they can produce the largest possible number of healthy offspring. These examples include eagle and pelicans. If food is abundant, then all eagle chicks in a nest will be well fed. If it happens to be a lean year, then the youngest, smallest chicks loose out, but the larger, older chicks survive. Pelicans produce two eggs each year, but only one chick ever survives to leave the nest. The second egg is nothing more than insurance against the possible failure of the first egg.
However, parent birds of most species do not begin incubation immediately after the first egg is laid. The parents wait until the last (or second-last) egg is produced. Because embryonic development does not begin until incubation is more or less continuous, all of the eggs in a nest will hatch at more or less the same time.
The advantages of synchronous hatching are many. Chicks in a nest are particularly vulnerable, and so it is in the parent’s best interest to keep that period as brief as possible. If all of the chicks in a nest hatch at the same time, none will have a competitive advantage over the others, increasing the probability that all will survive. If all chicks are at the same stage of development, then they will all be ready to leave the nest at the same time. This means that parents will not have to feed both chicks in the nest and chicks that are exploring the world. With all of these advantages, it is not surprising that birds of so many species start incubation when the last egg is produced.
However, there are exceptions. If we claim to understand the biology of birds, then those exceptions will require explanations. Next week I will describe how recent research has come to terms with the asynchronous hatching of chicks of an exceptional Australian finch.
Photo credits: Brown Pelican and chick, © Bryan Watts – www.ccbbirds.com; Bald eagle and chicks – animalli.com
Week 68 – 05 November 2017:
Small Birds and Pink Elephants
There are many urban myths associated with birds. One of my favourites is that, when given the opportunity, birds will gorge upon Yorkshire puddings to the point where they become too heavy to fly.
Stories about fruit-eating birds and mammals becoming drunk on the alcohol in decomposing fruit are not so much mythical as poorly-investigated. It seems that reports of the intoxication of elephants that have ingested overly-ripe fruit may have been a misinterpretation of the situation, related more to the ingestion of other compounds including nicotinic acid.
The potential for alcohol ingestion by birds certainly exists. After all, the whole point of a plant producing fruit is to have it consumed by an animal who will then disperse its seeds. The reward for the animal is the fruit’s energy-rich pulp. In order to attract a fruit-eating bird or mammal, fruits are generally brightly-coloured, and their smell is likely to be pleasant.
Could it be that fermenting fruit is even more attractive to frugivores that less-ripe fruit? Birds might discover fermented fruit after being attracted by the scent of alcohol. Furthermore, the caloric content of ethanol is particularly high. Frugivorous birds are bound to consume some alcohol, but is the alcohol particularly attractive?
Mannqoba Zungu and Colleen Downs of the University of KwaZulu-Natal in South Africa sought to determine whether birds consumed more food when it contained ethanol. Their study involved ten Cape White-eyes, ten Red-winged Starlings and six Speckled Mousebirds, all captives. Subjects were fed on an artificial diet suitable for fruit-eating birds. Red food colouring was added to the food to make it more appealing. Half of the food offered to the birds contained one percent ethanol, and the other half did not.
For members of all three species, there was no statistical difference in consumption of the experimental and control food, although there was a tendency toward consumption of food lacking ethanol. It seems that alcohol does not act as a positive foraging cue, at least in these species.
Zungu and Downs pointed out that the results of their study should be considered preliminary, and that inferences based on their data are modest. Despite the appeal of studying alcoholic fermentation, we know very little about the alcohol content of over-ripe fruit of most plant species under natural circumstances, particularly in the tropics. The study of Zungu and Downs exposed bird to food with only one concentration of alcohol. More alcohol or less might prove more appealing. Furthermore, it seems likely that birds of different species metabolize ethanol at different rates, based on whether they might experience alcohol frequently, as frugivores would, or rarely as granivores would. Further study is needed.
In the meantime, there is a bottle of pinot grigio chilling in the refrigerator.
Zungu, M. M. and C. T. Downs. 2017. Effects of ethanol on fruit selection by frugivorous birds. African Zoology 52:69-72.
Photo credits: Painting of Cape White-eyes by C. G. Finch-Davies - www.hathitrust.org; Red-winged Starlings – www.pinterest.com
Week 67 – 29 October 2017:
Tits in the Himalayas
A number of my colleagues study bats. Some of those researchers consider a remarkable phenomenon know as elevational species replacement. As one moves uphill from habitat at low elevation to high, a particular bat species will get replaced by a similar but different species of bat, and then by another. Birds, amphibians and reptiles all show this sort replacement of one species with another with changes in altitude.
There are two possible reason for this sort of replacement. It could be that each species is particularly-well adapted for the conditions found within a given altitudinal range, and that individuals remain in their preferred habitat. Alternatively, one species might be a particularly good competitor, forcing members of other species to accept more marginal habitat at a different altitude. There has been considerable study of elevational species replacement in tropical zones, but far less in more temperate regions.
Sahas Barve and André Dhondt of Cornell University in Ithaca New York sought to address that deficiency by studying two tit species in the Himalayas of India. Unlike the situation in the tropics, birds in the Himalayas are likely to face very large seasonal variation in temperature, and considerable differences in the availability of resources in the breeding and nonbreeding seasons.
The birds studied by Barve and Dhondt were the Green-backed Tit which breeds at higher altitudes, and the Cinereous Tit breeding at lower altitudes. They overlap at an altitude of between 1300 and 1400 metres. These hearty little birds are both year-round residents throughout the Himalayas, and are very closely related to one another. They occupy a broad range of habitats that include forests and human settlements. In regions in which the habitat has been altered by humans, including settlements and nearby agricultural lands, Cinereous Tits can be found at elevations as high as 1600 metres. What determines these patterns of distribution?
Barve and Dhondt studied the responses of tits to the playback of songs at three altitudes. One region was low enough to be occupied by Cinerous Tits alone, one was high enough to be occupied only by Green-backed Tits, and one was of intermediate altitude and had both species. At all three sites, individuals heard the playback of songs of their own species, and of the other species.
Not surprisingly, at all three sites, individuals of both species responded strongly to playback of songs of their own species by approaching the playback speaker, and responded less aggressively to the playback of songs of the other species. In the region where both species were found, Green-backed Tits were more likely respond aggressively to the playback of Cinereous Tit song than the other way around, and the researchers concluded that Green-backed Tits were the behaviourally-dominant species.
At the altitude at which both tits could be found, Barve and Dhondt provided feeders at which the birds might interact in the non-breeding season. They found that Green-backed Tits were more likely to displace Cinerous Tits from the feeding opportunity than the reverse situation. Again, Green-backed Tits were the behaviourally-dominant species.
Despite this dominance of Green-backed Tits over Cinereous Tits, it doesn’t seem that aggressive behaviour determines the altitudinal distribution of these two species. Given a significant overlap in distribution, it seems more likely that habitat preferences might be more important. Although Cinereous Tits are common in forested habitat at lower altitudes, they seem to be restricted to regions of human habitation in the regions where both species found. When I contacted Barve, he wrote to say: “I personally think cinereous tits live at those (mid) elevations purely because people regularly feed birds there. I also think they can live up there (because) they roost in people’s houses, in slots under the eves so they can keep warm on long cold winter nights.”
I asked Barve whether the residents of the Indian Himalayas are particularly keen on the tits in their communities. He felt that there was no special fondness for tits, and the locals do not have a special term for that sort of bird. Instead the tits get lumped together with other small perching birds under the title of “sparrows.” However, Barve indicated that: “It is considered a good thing in Hinduism to feed animals instead of throwing stale rice or old bread in the garbage,” and so “they just throw it in their backyard and the birds love it.”
Barve explained to me that fieldwork in that region can be challenging, but that when operating in a region surrounded by Himalayan peaks up to 7000 metres, and with 250 bird species to watch, “it is too much fun to hate any part.”
Barve, S., and A. A. Dhondt. 2017. Elevational replacement of two Himalayan titmice: interspecific competition or habitat preference? Journal of Avian Biology 48:1189-1194.
Photo credits: Green-backed Tit – www.pinterest.com; Cinereous Tit © Lip Kee – www.thainationalparks.com
Week 66 – 22 October 2017:
A Question of Colour
Last week, I wrote about the distinguished Cambridge University professor Alfred Newton (1829-1907). On 18 November, 1869 Newton published a paper in the journal Nature, describing the great variation in egg colouration among parasitic Common Cuckoos. He speculated that an individual female cuckoo might lay her eggs in the nests of a particular host species. If the colouration of the hosts’ eggs matched her own, they might avoid detection, and so be more likely to survive. Newton went on to suggest that over a period of years, the colouration of eggs, and the tendency of females to lay in the nests of particular hosts might become heritable traits.
I strongly suspect that these final thoughts about heredity were Newton’s attempts to tie the colour of cuckoo eggs to Charles Darwin’s ideas about evolution by natural selection. On the Origin of Species had been published just ten years before, and biologists of the day were still struggling to deal with the profound implications of Darwin’s ideas.
Not everyone was convinced that Newton had made the correct conclusions. Just two weeks after Newton’s 1869 paper, a response appeared in Nature, written by W. J. Sterland, author of The Birds of Sherwood Forest. “I have no intention to criticise so able and accomplished a naturalist,” wrote Sterland, but rather to “elicit information on some points of difficulty.” While Newton described remarkable variation in colouration of cuckoo eggs, it was the experience of Sterland and some of his colleagues that there are few species of birds whose eggs are less variable.
When it came to Newton’s ideas that a female cuckoo would lay her eggs in the nest of just a single species, that her eggs would resemble those of her hosts, and that the process might be heritable, Sterland had his greatest concerns. If sixteen varieties of cuckoos each produced a particular type of egg, each parasitizing a different host species, then “how are these sixteen varieties to be kept from crossing?” If members of the population do interbreed, “how can the alleged distinctive style of eggs be preserved?”
To be fair to Newton, no one in 1869 could fully answer that question. An understanding of the principles of the inheritance of characteristics had to wait more than thirty years for an appreciation of the research of Gregor Mendel on pea plants.
In a note published on December 23, the eminent ornithologist Henry Eeles Dresser (1838 – 1915) added his thoughts on Newton’s paper. Dresser was particularly interested in the breeding biology of brood parasites, and objected to the notion that cuckoo eggs were particularly variable in appearance. Unusually-coloured cuckoo eggs could be found, but he found them to be “of very rare occurrence.”
Dresser conceded that an individual female cuckoo might produced eggs that closely resembled each other, but that “I have not found that the same cuckow generally makes use of the same species of bird as nurse for her offspring.”
The following week, it was the turn of Cecil Smith (1826 – 1890), author of a number of books concerning the birds of Great Britain. Smith was not so polite in his criticism as Sterland or Dresser had been, perhaps because Newton had taken direct aim at Smith’s writings in the original cuckoo paper. Smith accused Newton of misreading the words of earlier writers, and manipulating them until “there is but little of the original left.” After three pages of critique of Newton, Smith went on to state that “Cuckoo’s eggs do not in fact vary from each other more than those of many others.”
The breeding biology of parasitic birds and their hosts remains a lively field of investigation. Studies of the arms race between them show the interactions to be beautiful shaped by evolution by natural selection. I hope that Newton would take some small comfort in that.
Sterland, W. J. 1869. Cuckows’ eggs. Nature 1:139.
Dresser, H. E. 1969. Cuckow’s Eggs. Nature 1:218.
Smith, C. 1869. Colouring of the cuckow’s egg. Nature 1:242-243.
Photo credits: Cover of the “The Birds of Sherwood Forest” by W. J. Sterland, www.amazon.com; front page of the “History of the Birds of Europe, Volume 1″ by Henry Eeles Dresser – auctions.bidsquare.com.
Week 65 – 15 October 2017:
The Colour of an Egg Shell
One of the most wonderful things about science is that the discipline is self-correcting. Science is not dogmatic. In the search for truth, every discovery is open to reexamination and reinterpretation. No matter now esteemed the voice, all proclamations must stand up to the scrutiny of others.
Consider the work of Alfred Newton (1829-1907), naturalist, anatomist and ornithologist. Newton was a professor at Cambridge University, and a fellow of the Royal Society. If ever there was someone whose opinion could be relied upon, it was Newton. And yet…
In 1869, a scholarly journal with the unimaginative name of Nature began publication. Each week, folks with novel ideas to share about the natural world would write letters to the journal’s editor, spelling out those ideas. Nature continues to this day as one of the world’s most distinguished scientific journals, and published papers are still described as “Letters.”
In the November 18 1869 issue of Nature, a letter from Alfred Newton was published concerning the colouration of eggs of the Common Cuckoo. “Scarcely any birds,” wrote Newton, “has so much occupied the attention, not merely of naturalists, but of people generally, as the Common Cuckow of Europe.” Newton reminded the reader that “in the experience of most persons,” Common Cuckoos did not build nests of their own but rather laid their eggs in nets of other species. If “most persons” found that Common Cuckoos were brood parasites, I am not sure what the others felt they had seen. Rest assured that Common Cuckoos do not build nests, and so are absolutely obliged to parasitize the breeding efforts of other birds.
Newton went on to say that: “It has long been notorious to oologists that the eggs of the Cuckow are subject to very great variety in colour, and that a large number of birds laying eggs of very different colours enjoy the doubtful advantage of acting as foster-parents to the young Cuckow.” Birds of different species lay eggs that differ in appearance. If the colour of the eggs of individual Common Cuckoos was similar to the eggs of her victims, it might aid in deceiving the host. The host might not notice the cuckoo egg amongst her own, and so may fail to discard it from the nest. Newton stopped short of saying that it was universal that the eggs of parasite and host matched, declaring the relationship to be “approximate.”
Given the wide range of colours and patterns of host eggs, how might a cuckoo achieve the deception? Even in the 1860s, few would suggest that a cuckoo could determine the colour of her eggs through a voluntary effort. Instead, Newton reasoned that a female cuckoo might generally place her eggs in the nests of hosts of a single species. For that female, this might happen year after year. Newton wrote that he was “not afraid of hazarding the supposition” that the creation of eggs of a particular colouration is likely to become a heritable trait, just as the tendency of depositing eggs in one host species or another is also likely to become a trait that could be passed along to future generations.
Does any of this sound a bit odd? Next week I will describe a number of responses to Newton’s letter in the pages of Nature.
Newton, A. 1869. Cuckow’s eggs. Nature 1:74-76.
Photo credits: Professor Alfred Newton – www.zoo.cam.ac.uk; Reed warbler feeding a Common cuckoo chick, photograph from the National Picture Library/Alany – www.bbc.com
Week 64 – 08 October 2017:
Could it be as Simple as That?
The study of birds has revealed fundamental truths in the grander world of biology. Much of what we know about population dynamics, predator-prey relationships, reproductive behaviours, animal nutrition, and even the relationship between parasites and their hosts are the result of studies of our feathered friends.
Birds serve as hosts to a very wide range of parasitic creatures, both on and within their bodies. From tapeworms to feather mites, roundworms to blowflies, flukes to fleas, birds are taken advantage of. Birds can also be parasites themselves, stealing some vital resources from members of other bird species. Some seabirds such as gulls and frigatebirds are known to be kleptoparasites, stealing food from smaller seabirds.
A great deal of research effort in recent decades had been directed at the phenomenon of avian brood parasitism, where the resource being stolen is parental care. Obligate brood parasitism describes the situation in which a type of bird does not construct a nest at all, relying absolutely on another bird species to build a nest, incubate it eggs, and feed its chicks.
The host of a brood parasite will almost always have diminished reproductive success. The parasite chick is likely to outcompete, or even kill outright, the chicks of the host. Therefore it is not surprising that studies of brood parasitism in birds have revealed many anti-parasite strategies. Some hosts are able to recognize the eggs of parasites, and either toss them from the nest or destroy the egg by pecking holes in the shell. If unable to do either of these, some hosts will abandon their current nest if it is found to contain a parasite egg, and begin the nesting process all over again.
Wouldn’t it be easier if potential hosts could simply assess the risk of parasitism at a number of sites, and choose to breed when the risk was lowest? Jere Tolvanen of the University of Oulu in northern Finland and his colleagues considered the possibility that hosts of Europe’s Common Cuckoo might make choices of exactly that sort, avoiding locations where cuckoos were perceived to be more common.
Tolvanen and his crew selected eight forest sites. Twenty species of forest songbirds were observed at these sites. Twelves, including Eurasian Blackbirds and Song Thrushes are not considered to be hosts for cuckoos. The other eights species are known to be vulnerable to parasitism by cuckoos. The latter group included Common Redstarts, Tree Pipits, Bramblings and Spotted Flycatchers.
In the first year of study, at four of the eight sites the research team broadcast the calls of cuckoos. Might this serves as a warning to potential hosts that their reproductive efforts were at risk? The other four sites did not receive the playback of cuckoo calls, and so served as a control. In the second year of study, the treatment of the sites was reversed; playback sites become silent, and silent sites received playback.
Among bird species that are not vulnerable to cuckoo parasitism, playback of calls did not result in a decline in abundance in the breeding season. This seem sensible; if there is no threat, there is no need to make a response. Among the bird species that are vulnerable to parasitism, an equally sensible response was made. Playback of cuckoo calls resulted in a decline in bird density. The simplest interpretation is that these birds considered the broadcast calls of cuckoos as evidence that their breeding season was at risk, and so decided to nest elsewhere.
Of the eight bird species that are known to be hosts to cuckoo parasitism, one did not respond as the others did. Although they are a frequent host of cuckoos, the breeding density of Common Redstarts did not differ between playback and silent sites. Tolvanen et al. speculated about why the redstarts did not respond to playback when other hosts species had. Common Redstarts are the only cavity-nesting species in the group, and it could be that appropriate cavities are so limited that any nesting site is too precious a commodity to pass up. Perhaps predation is a far greater risk to redstarts than brood parasitism, and so they dismiss the calls of cuckoos as unimportant. It might be that “Common Redstarts are either not capable of perceiving cuckoo vocalizations as an indication of high parasitism risk or do not use such information in habitat selection.” wrote Tolvanen et al.
Avian brood parasitism is an arms race in which parasites try to get better at their sneaky reproductive strategy, and hosts keep trying to avoid being taken advantage of. Many, many interesting studies will be conducted in the future.
Tolvanen, J., J. T. Forsman and R. L. Thompson. 2017. Reducing cuckoo parasitism risk via informed habitat choices. Auk 134:553-563.
Photo credits: Common Cuckoo stamp – www.ebay.com; Common Redstart – ArtMagenta.com
Painting of Common Redstart by ArtMagenta. (ArtMagenta.com)
Week 63 – 01 October 2017:
The Parakeet Fights Back
Birds can be endlessly attractive and engaging. Regrettably, being cute doesn’t protect birds from the bacteria, viruses, fungi and protists that infect members of all animal groups. Fortunately, when living at reasonably low densities in the wild, an otherwise-healthy bird is likely to be able to fight off most pathogens.
Once birds are taken from the wild and into captivity, and held in closer quarters, the story can change. The risk of transmission of communicable diseases can become much greater, not just to the birds themselves, but also to the humans that share their living spaces. Consequently it is necessary that zookeepers, pet store staff, poultry producers, quarantine staff, and pigeon fanciers monitor the birds in their care, watching for signs of ill-health.
As with other animals, an individual bird can serve as a host to viral and bacterial agents long before it shows any pathological signs of disease. An apparently healthy bird might be infective to those around it. It will, in some situations, be prudent to routinely screen for pathogens in a captive flock whose members all look healthy if later outbreaks are to be avoided. Swabs can be taken from the bird’s throat or its cloaca (the shared opening of the bird’s digestive, urinary and reproductive systems). Whatever was collected on the swab could then be cultured on a nutrient medium, or subjected to diagnostic genetic analysis. The difficulty is that sampling of this sort is invasive, time consuming, and potentially stressful on the animals being tested.
Alenka Dovč of the University of Ljubljana in Slovenia and thirteen of her colleagues investigated the possibility of utilizing noninvasive sampling techniques to monitor the health of captive birds. In particular, they studied the diagnostic value of filtering large samples of the air in bird enclosures.
In particular Dovč and her group studied the presence of airborne pathogens in four holding facilities for parrots. Although their paper did not say so specifically, contextual clues suggest that these were large-scale facilities that supplied parrots for the pet industry. Among the birds in these holding were Blue-throated Macaws (critically-endangered in the wild), Eclectus Parrots, Rose-billed Cockatoos, Orange-winged Amazons, and Great Green Macaws (endangered in the wild). In addition, Dovčet al. sampled the air from fifteen racing pigeon facilities. There are, presumably, a lot of pigeon enthusiasts in Slovenia. For comparative purposes, the research team also performed more traditional sampling.
The list of diseases that might be spread from one bird to another was, quite frankly, rather frightening. It included such diseases as psittacine beak and feather disease, and proventricular dilation wasting disease. Not all of the diseases would be restricted to bird hosts. Among the infections that have a high or moderate risk for humans are avian influenza virus, Chlamydia psittaci, Candida parapsilosis, giardiasis, salmonella, West Nile virus, cryptosporidiosis and cryptococcosis. Yikes!
Air sampling proved to be valuable in revealing infections of PDDV and CP, but less successful in the case of other infections such as psittacine beak and feather disease virus, adenoviruses and Mycobacterium avium complex. It seems that in avian health, as in human health, a comprehensive approach to diagnostics is likely to provide the best results.
How is your pet parakeet looking?
Dovč, A., et al. 2016. Occurrence of bacterial and viral pathogens in common and noninvasive diagnostic sampling from parrots and racing pigeons in Slovenia. Avian Diseases 60:487-492.
Photo credits: Blue-throated macaw artwork – www.pinterist.com; Rose-billed cockatoo (galah) artwork – www.redbubble.com
Week 62 – 24 September 2017:
Eggs For Supper? Again?
If you are not familiar with the foraging behaviour of Black Skimmers, Rynchops niger, I would like to suggest that you search for videos online. I will not spoil the surprise by describing that behaviour, but sincerely believe that you will not be disappointed.
Black Skimmers range from the southern United States and Mexico to Venezuela and Argentina, with a global population that is considered to be of least-concern, but in decline. Seabirds, these skimmers nest in modest-sized colonies in open situations, such as gravel and sandy beaches. While many seabirds nest on remote islands, presumably in order to avoid mammalian predators, Black Skimmers sometimes breed in less-isolated situations.
Elizabeth Forys of Eckert College in St. Petersburg and her colleagues observed the nesting of skimmers in Pinellas County on the west coast of Florida. Here the birds chose beaches that were lined with condominiums and hotels, and each day the birds shared their breeding habitat with hundreds of sun-seekers. Unfortunately, these Black Skimmers also had to share their world with Fish Crows, Corvus ossifragus.
Between 2005 and 2011, a colony of Black Skimmers had a degree of breeding success, fledging between 22 and 72 chicks each year. Things started going wrong for the skimmers in 2012, when they experienced complete breeding failure as a result of egg-predation by Fish Crows. Forys and her colleagues considered using aversion conditioning on the crows. In this scenario, some eggs would be treated with a chemical that would make crows ill, but would not kill them, training the predators to avoid eating skimmer eggs in the future. This sort of conditioning has been used with success in the past.
However, the sorts of chemical that will cause a crow to vomit require permits. While waiting for those permits to be processed, Forys and her crew were advised to try a different approach. Within a colony of 72 pairs of Black Skimmers, the research crew placed a number of models of dead crows. Some of the effigies were hung above the ground, and others were laid directly on the beach. Would these gruesome scarecrows deter hunting by Fish Crows, protecting the Black Skimmers’ eggs?
In a word, no. Forys et al. wrote: “After we placed the effigies, several crows flew to where they (the models) were placed and examined them, but quickly resumed their previous activities.” An egg was taken by a crow less than ten minutes after the crow models were installed, and it was not long before all of the eggs had been preyed upon. On the final day of the study, June 3, the skimmers abandoned the colony, and they did not attempt to breed again that year.
Fish Crows are not evil. Instead, they are clever birds that take advantage of foraging opportunities. The activities of a small number of crows on a single urbanized beach in Florida will not imperil the long-term survival of Black Skimmers. However, the work of Forys and her colleagues shows that some conservation questions do not have simple answers. The more we know about the natural history of all the players, the greater the likelihood that we can create solutions.
Forys, E. A., D. Hopkins, P. Ingham, M. Miller and L. Gluckman. 2015. Do effigies deter Fish Crows hunting in a Black Skimmer colony mid-season? Southeastern Naturalist 14:635-640.
Photo credits: adult and juvenile Black Skimmers, photograph © Laura Erickson – http://www.lauraerickson.com & www.flickr.com; Black Skimmers painting © Julianne Felton – fineartamerica.com
Week 61 – 10 September 2017:
Go Outside and Play!
There can be no doubt that our parents’ commands to get some exercise were exactly what we needed to hear. The development of our young bodies benefitted from the exercise, and our coordination likely improved too. Playing outside was good for us. Might that sort of early exercise also benefit young birds?
The precocial young of many types of birds, including ducks and shorebirds, are ready to run almost as soon as they hatch out of their eggs. For many other birds, altricial species, there is a prolonged period of growth and development before chicks are ready to leave the nest. As the date for departure approaches, young birds in the nest can be seen flapping their wings. This behaviour presumably aids in all-important growth and coordination required for survival after leaving the nest. The issue of crucial pre-fledging exercise is probably particularly acute for seabirds that nest at the ends of tunnels, where room to stretch is at a premium.
The Streaked Shearwater is a seabird that breeds on islands off the coasts of China and Japan. According to the International Union for the Conservation of Nature, it is near-threatened, and with a declining global population. In order to help avoid arrested development of their muscular and nervous systems, young Streaked Shearwaters are known to periodically emerge for brief excursions before returning to their nests. While outside the nests, the chicks were thought to exercise their wings. This behaviour is considered to be quite common among burrow-nesting seabirds, but it has been poorly studied, particularly since it almost always happens at night.
Ken Yoda and his colleagues at Nagaoka University and Nagaoka University of Technology in Japan studied the exercising behaviour of young Streaked Shearwaters in the period leading up to fledging. The study was conducted in 2013 and 2015 on Japan’s Awashima Island. Infrared video cameras placed in front of the entrances to shearwater nests documented the time at which chicks emerged from, and then returned to, their nests. Some chicks were also fitted with acceleration-temperature devices that logged the period that chicks spent flapping. Chicks were weighed, the length of their wings measured, and their ages estimated.
Of the twenty chicks studied, Yoda and his team documented 805 excursions. The average age of first emergence from the nest was 68 days after hatching; this was fifteen days before the typical chick fledged. Excursions averaged 32 minutes in duration, during which chicks were seen flapping their wings vigorously at a rate of four times per second for approximately six-and-a-half seconds.
Not every Streaked Shearwater chick behaved in exactly the same way. Yoda et al. discovered a positive relationship between the period that chicks spent exercising and their mass and wing length. More is better. Furthermore, chicks that exercised the most were able to leave the nest for the last time at a younger age.
The data presented a curious conundrum. “Our correlative evidence did not indicate whether high-quality growth promotes exercise or conversely whether exercise promotes high quality growth, but it is possible that both processes occur simultaneously.” Regardless of cause and effect, the study of Yoda and his coworkers indicated that the superior development of muscles and bones that accompanies exercise is likely to be linked to better survival of Streaked Shearwater chicks after they leave their nests.
So when you tell your children to go outside and play, you can feel entirely justified. Unlike Streaked Shearwater parents, you might even choose to spend time playing with your children.
Ken Yoda, et al. 2016. Preparation for flight: pre-fledging exercise time is correlated with growth and fledging age in burrow-nesting seabirds. Journal of Avian Biology 48: 881-886.
Photo credits: Streaked Shearwater (Calonectris leucomelas), © Robin Newlin – www.birdskorea.org; Streaked Shearwater stamp – www.hipstamp.com
Week 60 – 03 September 2017:
Is it Possible to be Too Popular?
I have never seen a Blue-crowned Laughingthrush, and there is a high probability that I never will.
According to the International Union for the Conservation of Nature (http://www.iucnredlist.org/details/22732350/0), this beautiful laughingthrush is critically-endangered. Its current population is unlikely to exceed 240 mature birds, all of which are found within an extremely restricted breeding range in China’s Jiangxi Province. It seems that, in the past, the trapping of individuals for the pet trade was the major cause of the bird’s decline, with 400 birds known to have been taken between 1987 and 1992. A subsequent international ban on the trade of Blue-crowned Laughingthrushes greatly diminished their value, and so the rate of capture declined. Even so, the species continues to be threatened by urban development and road construction.
A recent report by Weiwei Zhang and colleagues at the Jiangxi Agricultural University and the Northeast Forestry University described aspects of the Blue-crowned Laughingthrush’s natural history, and identified a new threat to its survival.
This laughingthrush has the misfortune to be both beautiful and rare. It is, therefore, popular with bird enthusiasts willing to travel. These birds breed only in broadleaf trees in and around a limited number of villages in the region of Jiangxi Province known as Wuyuan. The spot is considered to be among the most beautiful landscapes in China, and home to a rich diversity of birds. Among the nine breeding sites known to Weiwei Zhang and colleagues, the largest laughingthrush population is also the most popular with birdwatchers and photographers. Between 2012 and 2016, the research team found 82 nests at this site, Shimen. Other sites, such as Hexi and Zhongyun, had only one or two nests, and were not occupied in every year.
Weiwei Zhang and crew used a clever approach to determine differences in potential disturbance of nesting Blue-crowned Laughingthrushes by photographers; they counted the number of photographs of the bird posted online. The focused on the website Birdnet, which they described as “the main web site for bird photo sharing in China.”
The team discovered that most of the Blue-crowned Laughingthrush photographs had been taken in Shimen. Not only was that laughingthrush population the largest and best know, but it also provided room-and-board opportunities for visitors in search of the birds. The birds in Shimen built their nests higher in trees than they did at other sites, and shorter tree species that were used at other sites were avoided at Shimen. The birds also nested further from village buildings at the most disturbed site. The researchers concluded that “the disturbance from birding visitors may exacerbate the endangered status of this bird.”
There is no doubt that ecotourism can be a powerful tool in the struggle to conserve wildlife and wild spaces. A lot of people are willing to pay a lot of money for the opportunity to see new and exotic creatures. A tree, standing up, home to a beautiful bird, may be a lot more valuable to the local community than the same tree that has been cut down for lumber. Even so, it is abundantly clear that ecotourism cannot be allowed to endanger the wildlife that it is is featuring. Even though the Blue-crowned Laughingthrush is considered to be critically-endangered by the IUCN, it is not classified as a protected species in China. Consequently it not legally protected against excessive disturbance from birdwatchers and photographers.
I have never seen a Blue-crowned Laughingthrush, and for the sake of the bird, I probably won’t try to do so.
Weiwei Zhang, Jinze Shi, Huiqin Huang and Tao Liu. 2017. The impact of disturbance from photographers on the Blue-crowned Laughingthrush (Garrulax courtoisi). Avian Conservation and Ecology 12(1):15.
Photo credits: Blue-crowned Laughingthrush – www.pinterest.com; incubating adult by Phillip Edwards – www.hbw.com
Week 59 – 27 August 2017:
With Remarkable Insight
The first ever issue of The Ostrich was released in March of 1930. A publication of the South African Ornithological Society, it was to be the repository of important findings about birds in Africa. That first issue included a list of fifty-four persons who had indicated their intention to support the society. Among those persons were folks with such grand names as Commander Talbot Ponsonby, Colonel Sir Llewellyn Anderson and Captain Guy C. Shortridge.
Now in its 88th year, the latest issue of Ostrich contains papers including “Habitat preference of the eastern population of the Short-clawed Lark Certhilauda chuana in the Limpopo province, South Africa” and “Is the Black Harrier Circus maurus a specialist predator?” As important as these new publications are, I believe that there is also value in looking back to works from a bygone era.
Ambrose Austin Lane was one of those who indicated his support of the South African Ornithological Society in 1930. Born in Ireland in 1868, he was a farmer in the Potcherfstroom district of South Africa until his death in 1936. Lane was a nature enthusiast, and was a founding member of the South African Biological Society in 1916. Land also contributed a work to the first issue of The Ostrich entitled “Some notes on weavers and widow-birds.”
In that publication, Lane described aspects of the breeding biology of the Pink-billed Weaver, Quelea sanguinirostris. Modern guides do not recognize a species by that name, but it seems likely that Lane was referring to the Red-billed Quelea, Quelea quelea. He described the voices of thousands of these weavers as “reminding one of the rythmic (sic) cadence of some lively stream flowing swiftly over a pebbly course.” Lane also explained that the nests of these weavers were composed of long needles taken from Beefwood trees. The needles were not well suited for the task, as “the first strong breeze usually brought the flimsy structure to the ground.”
Lane also described the lives of birds now called Red Bishops, Euplectus orix. Cold weather caused Red Bishops to abandon their breeding grounds, and to “gather in flocks on the warmer part of the highveld… where they feed on the open ground, probably on grass and other seeds that are plentiful.” These sorts of observations were quite common in the early ornithological literature. Papers considered the natural history of birds, without adopting a scientific approach.
Lane’s 1930 publication was different; it went a step beyond the ordinary. He pointed out that South Africa was going through an agricultural revolution, as had already been seen in North America. Native habitats were being transformed into agricultural land. “Original species may be gradually exterminated and be replaced by quite different species,” Lane wrote, “which may lead to variations in type, habits or migratory range and would be interesting to follow.” He suggested that “respected members of the South African Ornithological Society by their cooperation can do much to throw light upon the life histories of birds under the changes that are taking place.”
Ambrose Austin Lane was, of course, completely correct. Observations made by many persons can reveal things that an individual or small group could not hope to. His words were also prophetic. The observations of many over a period of decades are starting to reveal changes in the lives of birds brought about by human-created global climate change.
Lane, A. A. 1930. Some notes on weavers and widow-birds. Ostrich 1:22-24.
Photo credits: Red-billed Quelea, © Lee Ouzman – www.astrocape.org.za; Red Bishop, © Steve Garvie – www.flickr.com
Week 58 – 20 August 2017:
XX, XY, ZZ and ZW
Male mammals make two kinds of sperm. Fertilization of an egg by one type of sperm, one that contains an X chromosome, result in daughters, while the other, with a Y chromosome, results in sons. Although the two types of sperm are made in exactly the same quantity, they are slightly different in their behaviour and longevity, and so mammals can influence the probability of bearing sons or daughters.
It isn’t quite so easy for birds. Male birds make only one type of sperm, and it is hen’s genetic contribution, Z or W, that determines whether an egg hatches into a male chick or a female chick. There is some evidence from a few species that birds may be able to influence the odds of having sons or daughters, but we don’t yet understand how that might happen. In most cases involving birds, the sex ratio at hatching is almost always 50:50.
That doesn’t mean that the sex ratio is necessarily 50:50 in the breeding population. In many types of birds, there is a male-bias, and that bias probably results from the additional predation pressures faced by females of some species as they incubate their eggs or brood their young.
A male-biased sex ratio in a population of birds takes on far greater significance if it occurs in an endangered species. Jonathan Kearvell and Megan Farley of New Zealand’s Department of Conservation studied the adult sex ratio in the Orange-fronted Parakeet, also known as Malherbe’s Parakeet. This critically-endangered parakeet is found in only three valleys on the mainland of New Zealand. In an effort to help ensure their long-term survival, individuals have been introduced to four small, predator-free islands. Although they are challenging to survey, a recent estimate put the mature parakeet population at between 150 and 200 birds.
Kearvell and Farley wanted to know if the adult sex ratio of Orange-fronted Parakeets was male-biased, whether this is a typical or abnormal situation for this species. They also sought to understand possible causes of a bias, and determine the influence of a bias on conservation programs. Although the observable differences between sexes are few, males can be distinguished from females on the basis of their slightly larger body size, their brighter colouration, and their longer bills. Between 2007 and 2012, observers tramped through forested valleys, identifying all parakeets to sex wherever possible.
In the breeding season, males constituted between 68% and 74% of Orange-fronted Parakeets observed. This bias isn’t too surprising, given that females remain inside the breeding cavity for prolonged periods. There was, however, a consistent bias in observations in the non-breeding season toward males, when they constituted approximately 58% of all parakeets seen. Kearvell and Farley attributed this inequity to predation of nesting females by introduced mammals. Plagues of introduced rats are particularly devastating for birds in New Zealand.
“The reduction in the number of females has an obvious conservation issue,” wrote Kearvell and Farley. “There are simply fewer mature females to produce the next generation.” Beyond the problem of numbers, an excess of unmated males can bring additional problems. These extra males can disrupt breeding by mated pairs, although the full effects of this interaction are currently poorly known.
Identifying a problem doesn’t necessarily provide an easy solution. In the case of critically-endangered Orange-fronted Parrots, programs to control non-native predators will certainly be an important step.
Kearvell, J. C., and M. R. Farley. 2016. Adult sex ratios in wild Orange-fronted Parakeets (Cyanoramphus malherbi): are there conservation implications? Notornis 63:26-31.
Photo credits: Orange-fronted Parakeet stamp – colnect.com; photograph – www.massey.ac.nz
Week 57 – 13 August 2017:
Many Mouths, All Well Fed
Advances in science do not always rely on expensive, sophisticated equipment. Sometimes the most interesting findings are the result of simple hard work. A recent publication by Martin and Pavel Čech of Charles University in Prague and the Czech Union for Nature Conservation typify this. The father and son team of Čech and Čech asked a comparatively simple question. How does the rate at which adult Common Kingfishers provide food to their chicks change with the number of chicks in the nest? 
The question might be simple, but the answer could be significant. An understanding of the food intake by piscivorous birds like kingfishers, herons and cormorants may have important implications for recreational and commercial angling, stocking rates, and even fish conservation programs. Given that each pair of Common Kingfishers will raise as many as four broods of young per year, each with between four and eight chicks, a lot of fish must be going into a lot of mouths.
Čech and Čech quantified the number and types of fish that Common Kingfishers took from rivers in Central Bohemia and the Highlands Region of the Czech Republic. Rather than attempting to determine kingfisher diet on the basis of video images, which may or may not be reliable, they chose a more direct approach. Common Kingfishers build their nests at the end of tunnels in riverbanks. The pair of investigators altered nest chambers, so that they could gain access from above. This enabled them to monitor the progress of chicks through the breeding season.
Čech and Čech were also able to take from the kingfisher nesting chambers all of undigested fish remains that had been regurgitated by the chicks as pellets. Such pellets are produced by chicks three or four times each day. The researchers washed and sieved these pellets to collect fish bones. These bones were were compared to a reference collection of bones to determine the size and species of fish. Amassing the reference collection, based on 776 individual fish, must have been a monumental task even before analysis of the kingfisher’s diet began.
From six kingfisher nesting cavities, 31,304 bones of 4,722 specimens of 24 fish species were retrieved. Among the most common of these were common dace, gudgeon, bleak and roach. The smallest fish delivered to a chick would have been 0.01 grams, and the largest 16.2 grams. Given that a Common Kingfisher weighs about 50 grams at the time it leaves the nest, the largest of the fish delivered must have been a challenge to consume. On average, each chick ate 37% of its own body mass each day.
From an ecological perspective, one of the most interesting results was that each chick in a larger brood was provisioned just as well as a chick in a much smaller brood. Being one of eight chicks in a nest proved to be no handicap.
When I contacted him, Martin Čech was able to further enlighten me about the challenging lives of Common Kingfishers in Europe. He explained that the bird’s nesting habitat, the bank walls of rivers and reservoirs, is being degraded as a result of the regulation of rivers. A wide assortment of predators are able to sense the nests, presumably by smell. These include otters, American mink, foxes, feral pigs and Norwegian rats. The artificial nests used by Čech and Čech diminish the threat of mammalian predators.
Winter also represents a big problem for Common Kingfishers in Central Europe. Some birds escape winter by flying to warmer climates, but some remain as residents. If the temperatures fall far enough to cover the rivers, then “they are facing death in a few days,” wrote Martin. He went on to say that some kingfishers exploit local up-wellings of warm water. Others resort to capturing strange prey in times of severe weather, as depicted.
Despite all of the challenges that are faced by Common Kingfishers, let’s hope that they continue to earn their name.
Čech, M., and P. Čech. 2017. Effect of brood size on food provisioning rate in Commmon Kingfishers Alcedo atthis. Ardea 105. doi:10.5253/arde.v105i1.a3.
Photo credits: Common Kingfisher on eggs – www.pinterest.com; Common Kingfisher with frog in winter, photographed by and © Pavel Čech.
Week 56 – 06 August 2017:
Evaluate, Then Reevaluate
All birds are beautiful. Some are more peculiar-looking than others, but that makes them all the more special. The Crested Ibis is just such a bird. With a red face, a mullet of long white feathers, and a ridiculously-long decurved bill, they make you want to take them home and feed them fresh muffins.
At one time, the Crested Ibis had a comparatively large breeding distribution in Russia, China and Japan. As a result of hunting, and destruction and pollution of their wetland habitat, the population fell and fell. In 1981, only seven wild individuals were known in central China.
But the news is not all bad. Although still considered to be endangered by the IUCN, the global population has now risen to something like 250 mature individuals. The best of their habitat in China’s Shaanxi Province is secure. In Japan, the village of Niibu-mura is home to the Sado Japanese Crested Ibis Conservation Centre, with captive-breeding facilities that may soon allow for releases into the wild. There is even talk about the possibility of establishing a wild population of ibis in South Korea. Of late, reproductive success has been quite high in both wild and captive populations. With that good news comes the opportunity to reevaluate management practices.
Flooded paddy fields and the wetlands that surround them are important habitat for Crested Ibis. They hunt there for fish, frogs, mollusks and aquatic insects. Winter is particularly challenging for these birds because their food is often in short supply. When research was conducted fifteen years ago, the single most important factor determining winter habitat use by Crested Ibis was the the size of paddy fields. Large fields from which one crop a year was harvested, followed by a long period of flooding, seemed to suit the birds best.
Since that study, the number of Crested Ibis, and the area occupied by the population, has expanded greatly. Can-Shi Hu of Beijing Forestry University and colleagues reexamined winter habitat use by Crested Ibis at the Hanzhong Crested Ibis National Nature Reserve in central China. Their methods were straightforward. They conducted field surveys in December 2011 and January 2012, and noted the habitat characteristics of used and unused paddy fields, reservoirs, mud flats and riverbanks. Binoculars, a motorbike and notebooks constituted their field gear.
Fifty-four sites were utilized by ibis, and fifty were not. Flooded paddy fields were still utilized, but mudflats and riverbanks had also become important sites for foraging. The size of paddy fields had proven important in the earlier study, but now the most important habitat features were the degree of human disturbance, the softness of the foraging substrate, and the distance to the nearest road. This sort of information can guide management practices in the decades to come.
Hu et al. wrote that for the sake of the Crested Ibis: “the local people should be encouraged and compensated to maintain single crop cultivation, plow paddy fields after harvesting, and irrigate them with shallow water in winter.” However, in recent years, many young people in Xangxian County have abandoned their farming income to become migrant workers. This migration has resulted in less of the habitat that is most desirable to Crested Ibis. Agricultural practices in the area are also shifting away from the type that results in ideal winter foraging habitat.
Even so, I remain optimistic. A little encouragement and compensation can go a long way.
Hu, C.-A., X. Song, C.-Q. Ding, Y.-X., Ye, B.-P. Quing and C. Wang. 2016. The size of winter-flooded paddy fields no longer limits the foraging habitat use of the endangered Crested Ibis (Nipponia nippon) in winter. Zoological Science 33:345-351.
Photo credits: Crested Ibis stamp – www.pinterest.com; Crested Ibis – www.chinauniquetours.com
Week 55 – 30 July 2017:
Red is Red, Right?
Earl Godfrey’s The Birds of Canada, revised edition, was published in 1986. It made me sad. Descriptions of the Yellow-shafted and Red-shafted flickers from the first edition (1966) had been replaced by a single description of the Northern Flicker. Having been shown that the red-shafted and yellow-shafted forms hybridize where the they come into contact, the two species were relegated to a single species with two distinctive colour variants. According to the Biological Species Concept, if two distinctive groups can hybridize to form viable offspring in nature, then they are members of the same species, whether I liked it or not.
The distinction between the two forms is in the name. The shafts of the flight feathers of Yellow-shafted Flickers are yellow. There are no prizes awarded for knowing that the shafts of the flight feathers of Red-shafted Flickers are red. Males of the former group have black moustaches, and males of the latter have red moustaches. The yellow-shafted form is found over much of eastern and central North America, and the red-shafted form is found in western parts of the continent.
Yellow carotenoid pigments give yellow-shafted feathers their colour. Metabolic derivatives of the same pigment, known as 4-keto carotenoids, give red-shafted feathers their characteristic colour. It has long been known that the distinction is based on genetic differences between the eastern and western forms.
But here comes the twist… A sizeable portion of Northern Flickers residing in eastern North America, hundreds or even thousands of kilometers from the hybrid zone, have some flight feathers with red or orange shafts. Even more curious was the observation that some eastern individuals would have different numbers of red feathers in successive years. For decades, ornithologists tied themselves in knots trying to figure out how this could be. Could some western genes have slipped into eastern populations? Was it possible that a gene responsible for red colouration turned on and off? Surely the red colour wasn’t the result of diet, because flickers eat mainly ants which don’t contain red pigments.
A recent publication by Jocelyn Hudon of the Royal Alberta Museum in Canada and her co-workers provided the solution to the puzzle. Utilizing red feathers taken from museum specimens that had been collected in eastern North America, Hudon and her crew subjected them a bewildering array of extraction and analytical techniques. It looks so impressive when people write things like: “The ether extract was evaporated over a stream of N2 gas and the pigments redissolved in a known volume of hexanes… using anhydrous sodium sulfate powder.”
What did Hudon et al. discover about flicker feathers? The unusual red colouration of feather shafts of eastern flickers was the result of the chemical rhodoxanthin, and not the 4-keto-carotenoids found in western populations. It is unlikely that the flickers synthesized this compound themselves, but rather acquired it in their diet. Hudon and her colleagues felt that the most likely source was the berries of Tatarian, Morrow’s and Bell’s honeysuckles. These plants are native to Asia, but were introduced to North America in the 18th century, and are now widespread.
On occasion, some Northern Flickers in eastern North America must add a few honeysuckle berries to their ant-based diet. Pigments in the berries are deposited in feathers, turning them red. Although it took some clever sleuthing to figure it out, the mystery of red feathers in yellow -feather populations wasn’t so mysterious after all.
Hudon, J. et al. 2017. Diet explains red flight feathers in Yellow-shafted Flickers in eastern North America. Auk 134:22-33.
Photo credits: Red-shafted Northern Flickers in flight – www.birdingisfun.com; Yellow-shafted Northern Flicker – animalia-life.club
Week 54 – 23 July 2017:
When Your Most Important Resource is a Hole
To a biologist, the Atlantic Forest of Brazil, Argentina and Paraguay is among the most enchanting places on the planet. In some parts of the forest, a single hectare of land may be home to more that 400 species of trees. Within the forest, among its myriad flowering plants, dwell uncounted types of mammal, amphibian, bird, and invertebrate animal. A very high proportion of those species dwell in the Atlantic Forest and nowhere else.
It was probably a pretty rosy place to live before humans started cutting it all down. Between forestry, agriculture and hunting, very little of the forest remains in its primordial state. Doesn’t that make the remaining segments all the more precious?
The Vinaceous-breasted Parrot is considered endangered by the IUCN. There are thought to be fewer than 2,000 mature individuals, with “a rapid and on-going population decline” probably resulting from “illegal nest poaching, habitat destruction and (its) persecution as a crop pest.” This parrot is an obligate cavity-nester, but its shares its home in Argentina with approximately 75 other cavity-nesting birds. An assortment of social insects and mammals also occupy cavities in trees in the region. As with so many other cavity-nesting birds, the Vinaceous-breasted Parrot doesn’t create its own cavity, but relies on excavators such as woodpeckers or wood decay. It appears that suitable cavities are rare, and this many limit the population of some species.
Eugenia Bonaparte and Kristina Cockle of the Instituto de Bio y Geociencias del NOA in Argentina sought to determine how much overlap there is between the types of cavities used by the Vinaceous-breasted Parrot and other cavity-dwellers. If conservation efforts are to be successful, it is best to know what sorts of cavities would most serve the endangered parrot, rather than one of its potential predators. Bonaparte and Cockle considered species overlap in the the timing of breeding and physical characteristics of cavities, and whether cavities are used by the same or different species in successive breeding attempts.
The study was conducted over ten breeding seasons in an area of mixed forest that included about 90% of the Argentine population of Vinaceous-breasted Parrots. The region contains unlogged forest, as well as pastures, cropland, and tree plantations. Beyond the parrots under study, cavities in the study area were used by birds such as Barn Owls, Saffron Toucanets, Barred Forest-falcons and White-eyed Parakeets, as well as opossums, bees and wasps.
Over the course of the study, Bonaparte and Cockle documented 78 tree cavities that were occupied by 153 nests and 16 roosts of birds, mammals and insects. The timing of breeding by the Vinaceous-breasted Parrot overlapped with almost all of the other cavity-nesting birds. Similarly, the characteristics of the trees and cavities used by the endangered parrots was similar to those used by most of the other birds, mammals and insects. Active nesting cavities were frequently reused by other species. Competition for this limited resource is likely.
In terms of the conservation and management of the endangered Vinaceous-breasted Parrot, it would have been nice if they had utilized cavities that were not attractive to their potential competitors. This wasn’t the case. Even though there was considerable overlap in the use of nest sites, Bonaparte and Cockle were able to identify characteristics of trees and cavities that could make them targets for special consideration. They suggested that the best situations are cavities between seven and 40 cm in diameter, at least 10 metres off the ground, in trees with a diameter of at least 55 cm. Given that a large proportion of parrot nests were found on farms, it will likely be prudent to work with farmers to help protect the most suitable trees.
Without those efforts, the Atlantic Forest might become a little bit less enchanting for the Vinaceous-breasted Parrot.
Bonaparte, E. B., and K. L. Cockle. 2017. Nest niche overlap among the endangered Vinaceous-breasted Parrot (Amazona vinacea) and sympatric cavity-nesting birds, mammals, and social insects in the subtropical Atlantic Forest, Argentina. Condor 119:58-72.
Photo credits: Vinaceous-breasted Parrots at nest, photograph © M. Lammertink , posted in an “Author Blog”, Auk and Condor, featuring Bonaparte and Cockle discussing their paper -americanornithologypubsblog.org/2017/01/18/cavity-nest-niche-of-the-endangered-vinaceous-breasted-parrot-sharing-a-limited-resource-among-birds-bees-and-opossums/; stamp featuring the Vinaceous-breasted Parrot – www.birdtheme.org
Week 53 – 16 July 2017:
The Positive Impact of Harvest
Buller’s Albatrosses breed on small islands off the coast of New Zealand. With a global population of 32,000 breeding pairs, they are considered by the IUCN to be near-threatened. Some estimates show that numbers of these beautiful albatrosses are increasing, perhaps by as much as 3% per year. In an age of population declines, this seems like pretty good news. A recent publication by Susan Waugh at the Museum of New Zealand Te Papa Tongarewa in Wellington and her colleagues suggests that Buller’s Albatross may be getting assistance from a peculiar source.
Like the Buller’s Albatross, the Sooty Shearwater is a near-threatened seabird, but with a much larger global population. There are 180 Sooty Shearwater colonies in New Zealand alone, supporting approximately 4.4 million breeding pairs. Shearwater chicks, known as tītī, are harvested by some Māori groups, including members of the Ngai Tahu people. The harvest is considered an important component of the local culture, and helps to maintain social cohesion. Recent estimates have the harvest as high as 400,000 tītī each year.
How are the two seabird stories linked?
Waugh et al. reported on their observations of Buller’s Albatross at a large breeding colony at Solander Island. Fifty nests were monitored on the remote and uninhabited island. They also reported on the movements of seventeen albatrosses that had been fitted with GPS loggers. With regards to Sooty Shearwaters, interviews were conducted with three muttonbirders – the name given to individuals that harvest tītī. The harvest occurs at night. Bodies are processed the next day, and the non-edible parts are discarded into the sea. The traditional Māori harvest results in discarded material of between 78 and 120 tonnes each year.
The GPS loggers demonstrated that the albatrosses flew very long distances in search of food for their chicks, particularly over the Foveaux Strait off the south coast of New Zealand’s South Island. Curiously, several of the albatrosses were tracked to regions close to muttonbirding sites, including one bird that visited five such sites. Furthermore, the remains of tītī were found at five albatross study nests. These items had been rejected by the young albatross, and included heads, feet and feathers. “It is likely that a greater portion of chicks were fed soft parts of tītī over the two-month harvest period in April and May,” wrote Waugh et al.
On a global basis, pelagic seabirds are among the most threatened of all avian groups. The harvest of young Sooty Shearwaters is recognized as having an important role in maintaining local Māori traditions. It may also have an important role in maintaining populations of Buller’s Albatross.
Waugh, S. M., T. A. Poupart, C. M. Miskelly, J.-C. Stahl and J. P. Y. Arnould. 2017. Human exploitation assisting a threatened species? The case of muttonbirders and Buller’s Albatross. PLOS One https://doi.org/10.1371/journal.pone.0175458.
Photo credits: Buller’s Albatross, Credit@Leoviaflickr – thejupital.com/hooked-on-the-albatross; Sooty Shearwater – colnect.com
Week 52 – 09 July 2017:
The Story is in the Details
The IUCN considers the Kentish Plover to be of least concern, with a global population estimated at between 290,000 and 460,000 mature individuals. These shorebirds can be found on lake shores, saltpans and sand dunes from Gibraltar to Guinea-Bissau, from Hungary to Hong Kong, and from Uzbekistan to the United Kingdom. Numbers are in decline in some parts of the plover’s range as a result of disturbance of their costal habitat, pollution of wetlands, and urbanization. Despite those population concerns, there are thousands of birds species in greater peril to keep conservation biologists awake at night. And that is the end of the plover story, right?
No so. Sometimes the story is in the details.
The eastern North Atlantic Ocean is home to a biogeographical region known as Macaronesia. The island groups that constitute this region include the Canary and Cape Verde islands, the Azores, and Madeira. These islands differ in geological age, and in the distance between each other and mainland Europe and Africa. The geographical isolation of the island groups does not necessarily result in protection of the plants and animals found there, and many endemic species are seriously threatened by the activities of humans. Among bird endemics are the Cape Verde Sparrow, Azores Bullfinch, Canary Island Kinglet, and the Trocaz Pigeon (Madeira).
Mohammed Almalki of Taif University in Saudi Arabia and his colleagues studied the genetic and structural differentiation of Kentish Plovers in Macaronesia, comparing them to continental populations. If the island populations were substantially different from one-another, it would argue in favor of special conservation and management efforts.
Adult plovers were captured, marked for individual identification with numbered metal legs bands, and the length of their wings and legs recorded. Before being released, blood samples were collected from the birds. Almalki et al. discovered that Kentish Plovers dwelling on each archipelago were genetically distinctive from others in the Macaronesia group. They also discovered moderate differences in the morphological traits measured; it seems likely that populations were showing adaption to differences in local conditions.
The researchers concluded that genetic and morphological differences among the Kentish Plovers found in different island groups might justify a review of the way that the species are managed. Although the groups are not sufficiently different to be classified as different species, “We suggest that each archipelago is better treated as an independent management unit,” they wrote. There is an urgency in the need to develop conservation actions, particularly where populations are increasingly threatened, such as the Canary Islands.
And just to show you that the smallest details can result in a good story, let me tell you about the scientific name of the Kentish Plover. The genus name, Charadrius, refers to a nocturnal waterbird mentioned by the writers Aristophanes and Aristotle, the sight of which was said to have cured jaundice. The species name, alexandrinus, is a Latin word referring to the Egyptian city of Alexandria.
Almalki, M., et al. 2017. Morphological and genetic differentiation among Kentish Plover Charadrius alexandrinus populations in Macaronesis. Ardeola 64:3-16.
Photo credits: Canary Island Kinglet – www.pbase.com; Kentish Plover male and chick, © Sushyue Liao – www.flickr.com
Week 51 – 02 July 2017:
The Leading Cause of Divorce Among Birds Is…
Mammals are a pretty scandalous lot. In most species, males and females come together only long enough for insemination, and then go their separate ways. In contrast, males and females of most species of birds form a long-lasting pair bond. Even when the current breeding attempt comes to an end, the pair may choose to mate with the same individual in subsequent attempts. It seems that familiarity with one’s mate is likely to increase the chances of reproducing successfully. Simple. And yet, divorce is surprisingly common in socially-monogamous birds. If it is advantageous to stay together, then what causes a pair to divorce?
Nataly Hidalgo Aranzamendi and her colleagues recently published a paper in which they investigated the possible causes of divorce in socially monogamous birds. In order to reduce the number of potentially confounding variables, they chose to study a species in which individuals remain paired and maintain their territories throughout the year, Australia’s Purple-crowned Fairy-wren. The breeding biology of these birds is pretty special; a mated pair may be aided by a number of subordinate males and females who help to raise the young. Most of these helpers are offspring from previous breeding attempts.
Hidalgo Aranzamendi et al. recognized three possible causes of divorce in fairy-wrens. First, an individual may be looking at better options – seeking either a better territory or a better partner. Second, if the mated pair is too closely related, an individual might seek to avoid the difficulties of inbreeding. Finally, it is possible that a divorce is not always voluntary.
The research was carried out over a period of ten years at the Mornington Wildlife Sanctuary in the Kimberly region of Western Australia. The site is recognized an an Important Bird Area by BirdLife International, and administered by the Australian Wildlife Conservancy. Three-hundred and seventeen breeding pairs were studied. These birds occupy territories along creeks and rivers. Coloured leg bands allowed the researchers to distinguish among individuals. Genetic testing screened for paternity, and incestuous matings were investigated with pedigrees.
Hidalgo Aranzamendi and her crew demonstrated that divorce is anything but simple in Purple-crowned Fairy-wrens. There was a high divorce rate among individuals that had paired with close relatives, and so divorce was a way of avoiding further incest. Females that left their mates often obtained mating opportunities on higher-quality territories. Infidelity, as demonstrated by offspring that were not the product of their social fathers, was also a predictor of divorce. In a few cases, territorial females were evicted by other females, showing that divorce is sometimes forced. What causes divorce in birds? Lots of things do.
How complex can the breeding behaviour of fairy-wrens be? The research team found that one female, socially-mated to her son, had produced offspring by copulating with a male on an adjacent territory. She divorced her son, and entered the territory of the male with whom she had previously copulated. That male divorced his current partner who was also his daughter. Could that possible be right?
I contacted Hidalgo Aranzamendi to check. Apparently I had interpreted the paper correctly. She described it as “an interesting example of the purple-crowned soap opera.” It seems that other species of Australian fairy-wrens have an even higher incidence of parentage by males other than the social fathers.
Hidalgo Aranzamendi described her feelings about field work to me. “Watching and following these birds and discovering their partnerships, new romances and infidelities, was a great adventure.” As a field biologist, I knew just what she meant. “I recall my best moments in the Kimberleys as those watching the most beautiful sunsets at the end of a long-day of work while drinking a cold beer in the company of great friends. Nothing can beat that feeling.”
Why would anyone want to do anything other than study birds?
Hidalgo Aranzamendi, N., M. L. Hall, S. A. Kingma, P. Sunnucks, and A. Peters. 2016. Incest avoidance, extrapair paternity, and territory quality drive divorce in a year-round territorial bird. Behavioural Ecology 27:1808-1819.
Photo credits: Purple-crowned Fairy-wrens, © Michelle L Hall – phys.org; Purple-crowned Fairy-wren – animalia-life.club
Week 50 – 25 June 2017:
Urban Myths About Divorce
When it comes to the behaviours of animals, two urban myths seem particularly persistent. The first is that non-human animals are aware of impending natural disasters long before humans are, and that by noticing changes in the behaviour of our pets, we can avoid catastrophes. Trust me – your budgie finds out about an earthquake when its perch starts shaking, at exactly the same time you do.
The second persistent myth is that birds of one species or another mate for life. Some perpetuators of this myth expand upon it, claiming that an individual that looses its mate will refuse to choose a new partner, no matter how long it lives. Swans are probably the most frequently cited examples. Again, you might want to trust me – birds divorce, and they get on with their lives if their partner dies.
There several reasons why a pair of individuals might remain mated in successive breeding attempts. If birds show fidelity to their breeding grounds, returning to more or less the same spot each year, chances are quite good that they will conveniently find their mates from the year before. In other birds, such as penguins, it can take a very long time to establish a solid pair bond with a mate. If an individual divorces its mate, it might have to miss an entire breeding season while working on a strong relationship with another singleton. There are also good reasons for divorce, including infidelity and failed breeding attempts.
Birds sometimes mate with the same individual in successive breeding seasons, and sometimes they divorce. But surely that begs the question of how we would even notice marital consistency and divorce among birds. I have seen a lot of swans, and they all look pretty much the same to me. The simplest way to address the question is through the observation of populations of birds that have had bands applied to their legs.
The Journal of Field Ornithology was formerly known as Bird-Banding. For more than eighty-five years, the journal has been publishing the results of scholarly research on the lives of birds. The eminent American ornithologist Margaret Morse Nice published an article in the first volume concerning divorce. It was a response to an unnamed author who had written: “probably the majority of passerine birds change mates for the second brood.” Is that true? In those songbirds that raise more than one set of offspring in a year, do individuals change partners between broods?
Nice surveyed the recently published literature for examples of banded birds that had either remained with the same partner for successive broods, or had chosen a new mate. She found evidence of seven pairs of birds, including Brown Thrashers and House Wrens that had changed mates for the second brood. In one case, a male bluebird had a different mate for his second brood, but switched back to his first partner for the third brood. In contrast, Nice found twenty examples from eleven species that had maintained their pair bond throughout the season. These included Chipping Sparrows, Song Sparrows, and Gray Catbirds. Although the sample size was small, Nice was able to conclude that songbirds do not ordinarily change partners within a season, as had been suggested.
That was the state of things in 1930. How do we see the world eighty-seven years later? See next week’s entry for an answer.
Nice, M. M. 1930. Do birds usually change mates for the second brood? Bird-banding 1:70-72.
Photo credits: Brown Thrasher – animalia-life.club, Gray Catbird – www.pinterest.com
Week 49 – 18 June 2017:
Profound Discoveries About Little-Known Birds
The grand diversity of life ensures that no bird enthusiast could ever become bored with the group. With more than ten thousand species occupying endless niches in countless habitat types, ornithologists always have something new to investigate. This wealth of research opportunities was emphasized by the March 2017 issue of the scholarly publication The Wilson Journal of Ornithology. Featuring 15 major articles and 14 short communications, the first six papers described birds that I had never heard of.
Forests of Central and South America continue to hold many surprises for bird biologists. In 1986, a new antbird, the Rondonia Bushbird, was discovered in an Amazonian forest in southern Brazil. Almost all aspects of the biology of this antbird remained undescribed until Thiago Costa of the Universidade de São Paulo in Brazil and coworkers started to fill in the gaps. Previously known from just a single female specimen, the male’s plumage was described in detail for the first time. Grey and black, it is, admittedly, kind of dull when compared to the rust-and-black female. These birds occupies dense unflooded forests, and may be able to tolerate a degree of habitat alteration by humans. Given that they are antbirds, it is not surprising that Rondonia Bushbirds were found to eat mainly ants.
There are seven recognized subspecies of the Tawny-throated Leaftosser, distributed widely across Central and South America. Many researchers believe that the species should be subdivided. Jacob Cooper and Andés Cuervo of Louisiana State University in the United States studied variation in the vocalizations of this leaftosser across its range. They found five distinctive song groups, and these corresponded to differences in other observable characteristics. According to Cooper et al., we should recognize five species in place of one, with the names Tawny-throated Leaftosser, Isthmian Leaftosser, Dusky Leaftosser, Andean Leaftosser and Guianan Leaftosser. Time will tell if the remainder of the ornithological community agrees with them.
The Hawaiian Crow, also known as the ‘Alalā, persists only as two captive populations; one at the Keauhou Bird Conservation Center on Hawai’i Island, and one at the Maui Bird Conservation Center on Maui. The goal is to breed them in captivity, and release the offspring in order to reestablish wild populations. Ann Tanimoto of the University of Hawai’i and her coworkers studied the vocal repertoire of captive ‘Alalā. They hoped that their data would serve to indicate the range of vocalizations in wild populations in the past, and allow future researchers to follow changes in those vocalizations in future wild populations. “These comparisons can provide a better understanding of the way environmental factors influence social learning in oscine birds,” observed Tanimoto et al.
Fruithunters are elusive thrushes, found only in montane habitat on the island of Borneo. As with so many tropical birds, their breeding biology has been poorly documented. Over a seven year period, Adam Mitchell of the University of Montana and his coworkers made observations of forty-two Fruithunter nests. Most nests contained two eggs, weighing a little over six grams each. Only females incubated the eggs and brooded chicks in the nest, but both parents fed nestlings. Chicks fledged after approximately 18 days. For thrushes of their size, Fruithunters had small clutches and long periods before fledging. Mitchell et al. speculated that this slow rate of breeding might allow for a longer lifespan for adults.
Despite being abundant and having a wide distribution in South America, the breeding biology of the Wren-like Rushbird has been poorly studied. Nicolás Chiaradia and his coworkers at the Universidad Nacional de Mar del Plata in Argentina considered these birds in the wetlands of Buenos Aires province. Life seems pretty tough for breeding Wren-like Rushbirds. Forty-three percent of the nests they discovered were abandoned during the nest-building stage. A further 38% were preyed upon, and 5% were destroyed. Only 14% of nesting attempts successfully fledged chicks. Late in the season, when the wetlands dried up, almost all active nests failed.
Blue Manakins living in the understory of forests in southwestern Brazil seem to have a slightly better time of it. The sexual displays of these tiny fruit-eating birds has been reasonably well studied, but their breeding biology has received a lot less attention. Paulo Zima and his coworkers at the Universidad Federal de São Carlos in Brazil studied Blue Manakins at thirty-six nests over two breeding seasons. Their shallow cup nests were built over water, and all contained two eggs. Young nestlings has dark grey downy feathers over dark red skin. The chicks were fed by the female parent only, and this may be related to their frugivorous diet; acquiring enough fruit to feed the chicks may not be very demanding, allowing for male desertion. Overall, 34% of nests successfully fledged chicks.
For many species of birds, our understanding of their lives is quite limited. Future generations of ornithologists will still find endless questions awaiting answers.
Photo credits: Tawny-throated Leaftosser © Joao Quental, Macaulay Library ML36928781 – macaulay.org; Blue Manakins – 985thejewel.com/video-long-line-of-suitors-for-female-blue-manakin/
Week 48 – 11 June 2017:
As Though Seabirds Weren’t Already Cool Enough…
Some of the most beautiful traits that we admire in birds have evolved under the influence of sexual selection. Preference for a particular trait by members of one sex can, over time, lead an elaboration of that characteristic in members of the other sex. The more exaggerated the trait, the more appealing the individual. A recent publication by Christy Wails of the University of New Brunswick and her coworkers details a fascinating, previously undescribed characteristic of a stunning seabird, the Crested Auklet. It seems that the bill plates of Crested Auklets fluoresce.
I consulted with Wails, now a Ph.D. student at Northern Illinois University. I confessed to her that I knew very little about bill plates. Christy explained that “bill plates are keratinaceous accessory plates that Crested Auklets produced around the onset of the breeding season. As chicks hatch… these plates become loose and they’re shed, leaving a dull bill through the non-breeding season.”
Crested, Least and Parakeet auklets were studied by Wails and crew at Gareloi Island in Alaska. There is a breeding colony of about 460,000 auklets on the island. Individual birds were briefly exposed to blue light. No part of Least and Parakeet auklets fluoresced, but the bright orange plates on the bill and around the mouths of all Crested Auklets fluoresced when exposed to blue light. It was something of a chance discovery that came about when the crew was stuck in camp as a result of rainy weather. Two members of the field crew had NightSea blue flashlights; the fluorescence was unexpected.
The Crested Auklet paper was a one-off for Wails, as she has moved on to studies of other seabirds in New Zealand. A few of her colleagues are interested in taking the study forward. It is likely to be a fruitful avenue for study.
Our understanding of the vision of birds is still rather limited. It is clear that many birds can see near-ultraviolet light, but most seabird probably don’t. The fluorescent light given off by the bills of Crested Auklets in the breeding season is something that they can almost certainly see, and may be a characteristic that is used in mate selection. Further research will be needed to determine whether the observed fluorescence is the result of a pigment, as Wails et al. suspect, or whether it is a result of physical characteristics of the bill plates playing tricks with the light. I suspect that an ornithologist with an appropriate blue light might have a fun and fruitful afternoon examining museum specimens of seabirds in one of the larger research collections in Britain or the United States.
Wails, C. N., E. D. Gruber, E. Slattery, L. Smith, and H. L. Major. 2017. Glowing in the light: fluorescence of bill plates in the Crested Auklet (Aethia cristatella). Wilson J. Ornithology 129:155-158.
Photo credits: Crested Auklet portrait © Mike Danzenbaker - avesphoto.com; Crested Auklet photograph of full bird – www.pinterest.com
Week 47 – 04 June 2017:
Birds That Share Your Neighbourhood
At the upcoming meetings of the American Ornithological Society in East Lansing, Michigan, one of the most important topics of conversation will be the ways in which birds are challenged by a world increasingly dominated by humans. There will be significant discussion of recent studies in the field of urban ornithology. Many bird species are well represented in towns and cities, and we are are starting to understand how urbanization influences bird abundance and distribution.
Less well studied is the influence of the urban environment on the construction of bird nests, and whether ectoparasites are more or less common in nests in cities. Given how much time and energy birds devote to building their nests, and how badly birds can be harmed by ectoparasites such as fleas and lice, this is an important gap in our knowledge.
James Reynolds and six of his colleagues at the University of Birmingham studied the nesting of Eurasian Blue Tits along urban gradients in 2014. These tits occupied nest boxes at thirty sites in Birmingham in England’s West Midlands. At the end of the breeding season, 131 nests were collected and disassembled. The component parts of the base and the lining of each nest were teased apart, and categorized as moss, grass, animal fibres such as fur, and materials that had been created by humans.
The most surprising outcome of the study was an apparent lack of influence of urban development on the nests of the tits that occupied Birmingham nest boxes. Nest bases were composed of moss, and they were lined by the other materials described above, but nests in different parts of the city were remarkably similar in their construction. Material that had been created by humans, such as plastic and dyed wool, was found in 73% of all nests, but the location of the nest along the urban gradient did not influence the amount of that material used.
The only ectoparasites recovered from the Blue Tit nests were fleas. Admittedly the flea population was pretty robust, averaging 184 fleas per nest. Flea abundance did not differ among nests in low, medium and high urbanization sites. The only factor that did influence parasite load was nest mass; heavier nests tended to have more fleas.
The results of a single study of a single species of bird cannot be used to paint too broad a picture. Reynolds and coworkers did not find compelling evidence of an impact of urbanization on the nesting of Blue Tits in Birmingham. That is not to say that significant impacts would not be found in other species in other urban centres. “We believe that research over the next few years will significantly improve our understanding of the breeding biology of a number of (city-dwelling bird) species and we encourage others in other large cities to respond to the challenge of working along their respective urban gradients.”
Stay tuned.
Reynolds, S. J. et al. 2016. Does the urban gradient influence the composition and ectoparasite load of nests of an urban bird species? Avian Biology Research 9:224-234.
Photo credits: bird nesting boxes – www.britishbirdlovers.co.uk; Blue tit photograph © David Chapman – www.saga.co.uk
Week 46 – 28 May 2017
With the Best of Intentions
It is comforting to know that the world is full of good people. These are the sort of folk who stop and offer help when they see you puzzling over a street map. Those same people will attempt to assist an ill or injured animal, perhaps by taking it to a veterinary practice or a wildlife rescue centre. This sort of behaviour isn’t about trying to save a species from extinction. It is an attempt to do the right thing for an individual animal in need.
The IUCN considers the Long-eared Owl to be of least conservation concern. Somewhere between 2,000,000 and 5,500,000 mature individuals are spread widely over Asia, Europe, North Africa and parts of North America. There is no reason to believe that they will fall to extinction anytime soon. Even so, there are those who believe that every individual is precious, and will attempt to assist a Long-eared Owl in distress.
Alessia Mariacher of the Istituto Zooprofilattico Sperimentale delle Regioni Lazio e Toscana and her colleagues collected data about admissions of Long-eared Owls to thirteen wildlife rescue centres in Italy. It was their hope that the results of the study would help to illuminate the sources of wildlife morbidity and mortality in the region.
Between 2014 and 2014, 391 live Long-eared Owls were taken into rescue centres for assistance. Almost sixty percent of these were adult birds, and most of those were admitted during the non-breeding season. Not surprisingly, young birds were mainly brought for assistance between April and June. Of those individuals that had suffered trauma from collisions, most were adults. They had likely been struck by automobiles or had hit fences or windows. A disturbingly large number of adults had been shot. Some had been poisoned, possibly through secondary exposure to rodent poisons. Most younger owls were healthy at the time of admission, having been found on the ground or on branches in wooded habitat. Although many of these young birds were almost certainly still under the care of their parents, some were admitted for care in a malnourished or injured state.
Despite the best efforts of personnel at the wildlife care centres, 64% of adults died while in care. This is not an unusual statistic. On a far happier note, 81% of young birds survived the experience. Mariacher and her colleagues reinforced an important message for those who find young birds, apparently abandoned. There are many reasons for a chick to leave its nest including the avoidance of nest parasites and potential predators, and a search for a cooler spot; it may not be abandoned at all. When it comes to owls, “these chicks still depend on their parents for food for the next few months, and may appear helpless by well-meaning passers-by who collect them” wrote Mariacher et al.
If we wanted to know more about the ultimate causes of owl mortality, it would undoubtedly be desirable to complete a comprehensive veterinary assessment of each casualty. This could include screening for poisons, and a study of diseases that might be acquired by birds while in care. Ideally release would be followed by monitoring of the health and longevity of the former patients. Given that the resources needed for this type of investigation is unlikely to made available any time soon, we will have to learn what we can from studies like the one by of Mariacher and her colleagues.
Mariacher, A., R. Gherardi, M. Mastrorilli and D. Melini. 2016. Causes of admissions and outcomes of Long-eared Owl (Asio otus) in wildlife rescue centres in Italy from 2010 to 2014. Avian Biology Research 9:282-286.
Photo credits: Long-eared Owl stamp – www.pinterest.com; Long-eared Owl adult and chick, © Rick and Nora Bowers/VIREO – www.audubon.org
Week 45 – 21 May 2017:
Crows As Unwelcome Guests
Dar es Salaam may be the most interesting city in the world that I knew absolutely nothing about. A bit of reading quickly alleviated my ignorance. With about 4 million residents, Dar es Salaam is the largest city in Tanzania. It also has one of the fastest rates of growth in the world, and is likely to reach 5 million by the end of the decade. By the end of the century, it may be home to as many as 76 million people, making it one of the largest cities on Earth.
And then there are the House Crows.
In order to highlight the significance of introduced species, the IUCN Invasive Species Specialist Group constructed a list of 100 of the world’s worst invasive alien species. These are species that have taken up residence far from their native range because of the actions of humans. The list includes such obvious invaders as crazy ants, goats, brown tree snakes, African tulip trees and Dutch elm disease. Included on the list are three birds; the European Starling, Red-vented Bulbul, and Common Myna. After what I read recently, I can hardly believe that House Crows didn’t make the list.
House Crows are native to thirteen countries, including China, Myanmar, Nepal and Sri Lanka. Curiously, the species has also been introduced to twenty-six countries, including Bahrain, Israel, Malaysia, Barbados, and Kuwait. The Australian Department of Agriculture and Food considers the House Crow to be an “extreme threat.” It seems that these birds often arrive in foreign lands on boats.
In the Tanzanian city of Dar es Salaam alone, there are about 800,000 House Crows, descendants of individuals that were introduced to Zanzibar 125 years ago in the hopes that they would eat garbage. If they had stayed on Zanzibar garbage dumps, there wouldn’t have been a problem.
An introduced species isn’t necessarily an invasive species. In order to be considered invasive, a species must have a negative impact on native wildlife or on humans or the economy. According to a recent publication by Moses Shimba of the University of Dodoma and Frederick Jonah of the University of Cape Coast, House Crows in Dar es Salaam certainly meet the criteria of “invasive.” Shimba and Jonah pointed out that these crows have a devastating impact on native wildlife, eating native birds, fish, frogs, lizards and mammals. They strip orchards of their fruit, and eat young domestic poultry. The birds unashamedly enter homes and restaurants to steal food, and are known to spread disease bacteria such as Salmonella and E. coli.
Knowledge is power, and so Shimba and Jonah studied the nesting ecology of House Crows in Dar es Salaam. Which types of trees were selected as nesting sites? Where trees of particular heights and shapes preferred over others? The results might have implications for the control of invasive crow populations.
Ninety-three trees of six species that were measured in neighbourhoods that were recognized as having particularly dense House Crow populations. Of those trees, 47 housed 55 nests, which tended to be located in trees taller than ten metres. This may be a result of efforts to eradicate the crows; the birds may have learned that nesting higher meant greater protection from persecution. Trees with larger crowns were preferred for nesting, but did not always resulting in the greatest nesting success. Crows were most successful when nesting in mango, madras thorn and a type of flame tree, all introduced tree species.
Regrettably, there was no single factor that made for good House Crow nesting habitat. Shimba and Jonah recommended further study in the hopes that it “will help to reveal the still unknown causes of variation in nest success and help to provide useful information for the on-going population control and eradication programs of the crow.”
Remember not to park your car under a House Crow roosting tree when you next visit Tanzania.
Shimba, M. S. and F. E. Jonah. 2017. Nest success of the Indian House Crow Corvus splendens: an urban invasive bird species in Dar es Salaam, Tanzania.
Photo Credits: both images were obtained from www.pinterist.com
Week 44 – 14 May 2017:
In Which Shrub Shall I Build My Nest?
University student who have studied conservation biology under me know my mantra. “An original, intact, operating ecosystem without any introduced species cannot be improved upon.” Whatever was in place before humans started messing things up is best. That is all perfectly good, but finding such a system, untouched by people, is a challenge. For instance, virtually every place on Earth has been affected by plants and animals that have been introduced by humans. Scientific studies of the impact of these introductions are often initiated after the invader has become established, and a complete before-and-after picture of the habitat is generally lacking.
Thank goodness, then, for a group of ornithologists at the University of British Columbia who have been studying Song Sparrows on Mandarte Island continuously since 1975. Mandarte is a six hectare island off the west coast of Canada. It hosts a community of maritime meadow plants, and is home to endangered Garry oak. It also harbours an abundance of Song Sparrows which are year-round residents. The island is a workable size, and so researchers have been able to band all of the sparrows for individual identification, and follow the progress of all of their nesting attempts. Song sparrow nest predation on the island is low, where almost all individuals nest in or next to a shrub.
Two shrubby species colonized Mandarte after 1963. They are the Himalayan blackberry, an invasive species from Eurasia, and the red elderberry, which is a native to the west coast of North America but also recognized as an aggressive invader when it colonizes a new spot. Using the long-term data available from Mandarte, Merle Crombe and his associates studied the responses of Song Sparrows to Himalayan blackberry and red elderberry.
The three most common native shrubs on Mandarte are snowberry, Nootka rose, and trailing blackberry. Coverage of these three species declined from 92% in 1986 to 58% in 2006, with the increase of the two colonizers. How did the Song Sparrows react to this change? They showed a strong preference to nest in the shrubby trailing blackberry (native) and Himalayan blackberry (introduced). The sparrows nested in the native Nootka rose and snowberry shrubs about as often as would be expected from their abundance. Of the 1051 sparrow nests in the dataset, only one was found in red elderberry, the regional native but recent colonizer; that nest failed. Excluding the single elderberry nest, the reproductive performance of Song Sparrows nesting in the other shrubs was similar.
The field of management of introduced species is incredibly complicated. Decisions about whether attempt to control or eradicate an invasive can only be based on our best guess of the costs and benefits of those actions. In the case of the Mandarte Island population of Song Sparrows, eradication of the red elderberry would probably be a good thing. This assumes that the removal of elderberry would enable natives to reestablish themselves, rather than be replaced by some other undesirable plant. It seems that eradication of Himalayan blackberry from Mandarte would make no difference to the resident Song Sparrows, but it could help to restore native plant species that are typical of the Gary oak and maritime meadow habitat which is, itself, endangered.
Crombie, M. D., R. R. Germain and P. Arcese. 2017. Nest-site preference and reproductive performance of Song Sparrows (Melospiza melodia) in historically extant and colonist shrub species. Can. J. Zool. 95:115-121.
Photo credits: Song Sparrows on Mandarte Island, © Martina Andrés – mandarte-island.weebly.com; Song Sparrow print by Walter Weber – www.allposters.com
Week 43 – 7 May 2017:
Peck, Peck, Peck
There was a time when ministers would include examples of animal behavior in their sermons in order to show parishioners a proper way to behave. See how the lioness protects her cubs against all threats, even at the risk of her own life. Behold the robin that continues to brood her chicks through the worst of storms. What a mistake that was! We now realize that in the struggle to survive and reproduce, animals are willing to engage in some pretty awful behaviours, none of which should be used as a model for human actions.
For example, while parents of most bird species are fully dedicated to the task, members of other species choose to shunt the responsibilities of parenthood onto others. An individual that lays its eggs in the nest of another bird is described as a brood parasite. A facultative brood parasite can, and probably will, make a nest and care for some of its own young, but will attempt to leave an additional egg or two in the nest of a neighbour. Far more sophisticated is the obligate brood parasite who has no intention of ever building a nest, relying completely on the nests of members of other species in which to lay their eggs.
- Natalia Cossa and her colleagues at Niversidad de Buenos Aires studied the behaviours of two species of obligate brood parasite in Argentina. As parasites, Shiny Cowbirds are generalists, known to lay their eggs in the nests of over 250 bird species. In contrast, Screaming Cowbirds are specialists, parasitizing only Baywings over much of their range.
As with many other species of brood parasite, when these cowbirds come to a host nest, they peck and puncture one or more eggs of their host. Cossa and her coworkers sought to determine why the birds would do this. They considered the possibility that if the host’s clutch was already completed, the hosts would hatch before the cowbird chick and outcompete it. There would be little point in a cowbird laying a parasitic egg if there was little chance that it would survive. If that is the case, then cowbirds might puncture the host’s eggs to force it to renest, giving the cowbird and opportunity to lay in the new nest.
Alternatively, could the cowbird be seeking to reduce the competition? By puncturing one or two host eggs, a cowbird might ensure that there would be abundant food for its own chick.
The study involved female Shiny and Screaming cowbirds taken into captivity. They were presented with nests with either one egg, implying that the clutch was not yet complete, or four eggs, implying that the clutch was already complete. Some presentation nests contained small eggs from House Wrens, others with larger and most robust Chalk-browed Mockingbird eggs, or the eggs of Shiny Cowbirds which were of intermediate size but were even more puncture resistant.
Cossa et al. found that eggs were equally likely to be punctured in completed clutches as in incomplete clutches, suggesting that the goal of the cowbird was not to cause the host to abandon its efforts and renest. When the nest contained four eggs, indicative of a completed clutch, the parasitic female did not destroy all of the host’s eggs. The researchers felt that this behaviour was most consistent with the idea that these cuckoos puncture eggs in order to reduce competition for their own chicks.
Birds may be beautiful beyond compare, but they can also be devious.
Cossa, N. A., D. T. Tuero, J. C. Reboreda and V. D. Fiorini. 2017. Egg pecking and puncturing behaviors in Shiny and Screaming cowbirds: effects of eggshell strength and degree of clutch completion. Behav. Ecol. Sociobiol. 71:60.
Photo credits: Shiny cowbird chick being raised by a Rufous-collared Sparow, © Nick Athanas – www.hbw.com; Screaming cowbird © Cesar Dario Lins – www.flickr.com & neotropical.birds.cornell.edu
Week 42 – 30 April 2017:
The Tenacity of Pied Bush Chats
In birds, parentage represents a huge commitment. There are a few hyper-precocial species in which chicks are fully able to care for themselves soon after hatching, but even in those species the effort required to get to that stage is enormous. As in humans, altricial birds are born nearly helpless, and rely on their parents for an extended period for food, warmth and protection. In balancing the demands of the chicks against the energy of the attending parents, is there a single best solution? Should parents always invest maximally in the welfare of their chicks, or might their efforts vary over time?
Navjeevan Dadwal and Dinesh Bhatt of Gurukula Kangrii University in India investigated changes in parental investment in the Pied Bush Chat. These small, altricial songbirds breed in southeast and central Asia and on the Indian subcontinent. As the name implies, males of this species are black and white, and fifteen subspecies are recognized across its vast range. The IUCN considers the Pied Bush Chat to be of least concern and with a stable population.
Dadwal and Bhatt examined the breeding biology of a population of Pied Bush Chats in the foothills of the Himalayas in northern India. Males were colour-banded for individual identification, and behaviours such as courtship, nest-building, incubating and provisioning were documented. The researchers asked whether males or females contributed more to nest defense, and whether the intensity of those defensive efforts changed as the breeding season progressed.
When a model of a predator, a stuffed Tawny Eagle, was placed near the nest, parent chats responded by giving alarm and threat calls, with males vocalizing significantly more. The authors speculated that females may have been in a weakened state after building the nests and laying the eggs. Alternatively, males may have been more willing to display as a way of advertising their quality.
As the incubation of eggs and the brooding of young progressed, chat parents increased their aggressive responses to the model predator, showing nearly four times as much response while caring for older chicks than for young eggs. As exposure to the model never resulted in a loss of chicks or eggs, perhaps repeated exposure caused positive reinforcement, such that the parents considered their defense efforts to be successful, and so responded more vigorously in successive tests. Given that Pied Bush Chats breed seasonally, parents may have been more inclined to defend more vigorously later in the sequence because less opportunity remained for a second attempt if the first attempt was defeated by a predator.
As an alternative explanation for the seasonal increase in defense behaviour, it could be that older chicks are more valuable to the parents than younger chicks, and that both are more valuable than eggs. Future costs to the parents of raising older chicks to independence are smaller than the same costs for young chicks or eggs, making them more valuable in the struggle by parents to pass along their genes to future generations. In any case it is nice to know that young Pied Bush Chats receive such good care.
When I wrote to Dadwall, he told me about some of the rewards involved with working with birds in his part of the world. “Studies of breeding ecology of Himalayan avifauna offer enormous possibilities for researchers, and certainly provide an excellent opportunity for insights into processes of diversification.” For instance, playback of warbler songs has illuminated the way that reproductive isolation can cause one species to diverge into two. “Such studies lead us to believe that the knowledge about bioacoustics and distribution patterns of particular taxa can significantly contribute to our understanding of the process of diversification.”
Once again, birds lead the way in providing answers to the great questions about nature.
Dadwal, N., and D. Bhatt. 2017. Examination of parental investment in nest defence in a tropical songbird, the Pied Bush Chat (Saxicola caprata). Avian Biology Research 10:19-23.
Photo credits: Male Pied Bush Chat, © Ron Knight - www.flickr.com/photos/9919745@N03/8079428108; Female and male Pied Bush Chats, © Brinda – brindays.wordpress.com.
Note, Ron Knight’s beautiful photograph is available via commons.wikimedia.org, attribution Ron Knight from Seaford, East Sussex, United Kingdom, Pied Bush Chat (Saxicola caprata) (8079428108), CC BY 2.0 .
Week 41 – 23 April 2017:
A Tough Time For Dippers
It is now well established that the activities of humans are resulting in significant global climate change. In particular, extreme weather events such as heatwaves, droughts and floods are becoming more severe and more common. How far this change is likely to proceed, and quite how fast, remain to be seen. Wildlife will certainly feel the impact of climate change, but studies of these consequences are needed desperately.
Brown Dippers are almost ideally suited for studies of the impact of extreme flooding. They are found throughout mountainous regions in Japan, Vietnam, Taiwan, China and other parts of central Asia. Pairs establish territories along fast-flowing streams and rivers, and feeds on large, aquatic invertebrate animals that they capture while walking underwater along the streambed. These dippers are comparatively common in places, and are not particularly secretive. They nest on cliff ledges, under bridges, and in cavities.
Shiao-Yu Hong of the National Pingtung University of Science and Technology and his colleagues investigated the impact of extreme flooding events on the reproductive performance of Brown Dippers. Their comprehensive study was conducted in streams of the Tachia River system in central Taiwan between 2003 and 2014. This period saw fifteen flooding events, varying in intensity and lasting from one to three days.
In Taiwan, the typhoon season typically runs from June to October. Hong et al. documented temperature and water flow. Brown Dippers were counted, and reproductive attributes such as laying date and nesting success were recorded. Further, the team sampled the identity and abundance of invertebrate animals in the streams in which the dippers foraged.
Floods resulted in a reduced availability of food for the Brown Dipper, and extreme flooding caused a decline in their survival. More insidious was the impact of flooding on reproduction. The breeding season of these dippers in January to April, several months after the end of the typhoon season. Even so, low food availability following severe floods resulted in later breeding, a lesser number of offspring produced, and a smaller dipper population the following year. For the birds, the most important feature was not the severity of each flood, but the length of time that had passed since the flood had ended. More time allowed the invertebrate animal population to recover.
It is not likely that the Brown Dipper will fall to extinction in the coming decades because of global climate change. However, increases in the frequency and severity of flooding that results from typhoons will almost certainly have negative consequences for them. The results of this study are an important indicator of how changes in climate will influence other wildlife species.
Hong, S.-Y., B. A. Walther, M.-C. Chiu, M.-H. Kuo and Y.-H. Sun. 2016. Length of the recovery period after extreme flooding is more important than flood magnitude in influencing reproductive output of Brown Dippers (Cinclus pallasii) in Taiwan. Condor 118:640-654.
Photo credits: Brown dipper in water, www.chinauniquetour.com; Brown Dipper family, www.pinterest.com
Week 40 – 16 April 2017:
Birds? What Birds?
Scrolling through very old issues of journals devoted to the lives of birds can provide remarkable insight into the ways in which the study of birds has changed. Building on decades of study, research today tends to ask questions that are far more specific than in the past. A century ago, ornithologists were concerned about fundamental aspects of the natural history of birds, rather than minor details of their genetic construction. However, a 1917 issue of the Wilson Bulletin, now known as the Wilson Journal of Ornithology, provides the only article about birds that I have ever come across that doesn’t mention even a single type of bird.
American ornithologist Frank Smith was born in 1857. He was granted a Bachelor of Philosophy degree by Hillsdale College in 1885 and an Artium Magister by Harvard University in 1893. He took up a position at the University of Illinois in Champaign-Urbana, where he was engaged for the remainder of his life. As both a professor in the university’s zoology department and a curator at its natural history museum, he doubtlessly influenced countless young lives.
Smith’s 1917 article in the Wilson Bulletin was, however, something of an oddity. The article’s title promised to demonstrate a link between the weather and spring migratory behaviour of birds. The data set was substantial, having been collected between 1903 and 1916. He explained that birds had been observed in an 18 acre artificial forest, and in Mount Hope Cemetery, immediately adjacent to the university campus. Students in Smith’s ornithology classes, employees of the university, and citizens of the city kept daily records of birds each year between early February and late May.
Smith considered the dates on which bird species were first seen in the spring, referring to them as “firsts.” “The data of “firsts”,” he wrote, “shows a great lack of uniformity of distribution throughout the season. The average season has had 89 days and the number of “firsts” has averaged 120 but these have been so bunched that 61 of them have been recorded on 9 days.” If you understand what any of that means, please tell me.
The days on which bird species were first seen in the spring was compared to daily weather maps of the region. “An examination of the weather maps for the 63 days on which are recorded a half of all the “firsts” of the last 30 days of each of the 14 season shows that on 54 of those days there were approaching areas of low pressure, with south winds which had been effective during the preceding night.” I think that means that southerly winds blow bird north. Smith went on to write that making graphs that compared temperature and wind direction with migratory activity would likely reveal some correlations. Regrettably he didn’t make any.
Let me reassure you that Illinois does, indeed, have birds. In fact 447 species are known from the state, eleven of which have been introduced, and two of which are extinct. Ross’s Geese, Pied-billed Grebes, Northern Harriers and Horned Larks can all be found there. The state bird of Illinois is the Northern Cardinal. You just wouldn’t know any of that from reading Smith’s article.
Smith, F. 1917. The correlation between the migratory flights of birds and certain accompanying meteorlogical (sic) conditions. Wilson Bulletin 29:32-35.
Photo credits: Northern Cardinal – www.pinterest.com; family of Pied-billed grebes, © Rolf Nussbaumer (www.rolfnussbaumer.com) from www.audubon.org
Week 39 – 09 April 2017:
More Discoveries About the Behavioural Ecology of Birds
In the last entry of A Traveller’s Guide to Feathers, I briefly summarized some recent discoveries concerning the behaviour of birds published in an issue of the journal Behavioral Ecology. Of the thirty-nine article in the issue, four were about fish, seven considered mammals, one was about reptiles, and three examined amphibian behaviour. Fully thirteen papers were about birds, demonstrating the importance of birds to our understanding of the world.
Birds of some species are less likely to establish territories close to roads, and several specific causes of this behaviour have been put forward. Matthew Schepers and Darren Proppo (Calvin University, USA) were able to increase the number of territories of six birds in Michigan by playing recorded bird songs near roads. These birds included the Black-throated Green Warbler and the Rose-breasted Grosbeak. These finding may have conservation implications.
Alex Grendelmeier (Swiss Ornithological Institute) and his colleagues also used the playback of recorded songs to investigate bird behaviour. They found that Wood Warblers in northern Switzerland were attracted to a site in which the songs of their species was broadcast. Regrettably some measures of the birds’ breeding biology was reduced at the site with playback, calling into question the value of acoustic attraction in songbird conservation.
Could it be that female birds alter the sex ratio of their chicks, producing more of the sex that suits current conditions? Male Stitchbirds of New Zealand rely on carotenoids to produce brightly-coloured feathers to acquire territories and attract mates. Kirsty MacLeod (University of Cambridge, UK) and her colleagues found that additional carotenoids in the diet of female Stitchbirds did not cause them to lay a greater proportion of eggs that produced sons.
Michelle Hall (University of Melbourne, Australia) and her co-workers studied the territorial responses of male Superb Fairy-wrens to the playback of recorded songs with trills of different lengths. Longer songs are perceived as being more threatening. The researchers were unable to find differences in the reactions of either bold or shy individuals to songs of different threat level.
Some birds, including the Blue-backed Grassquit of South America, perform displays that appear to be both energetically demanding and difficult to complete. Lilian Manica (Universidade de Brasilia) and her colleagues found that the characteristics of the grassquit’s display that require vigour, and those characteristics that require skill, may limit each other, and that signallers of lower quality are particularly impaired.
Alessanra Costanzo (University of Milan, Italy) and her co-workers found that Barn Swallow parents provided more care to chicks that were more darkly-coloured. Dark-colouration was also positively related to the length of telomeres, an attribute of the cell’s chromosomes that indicates high quality. It seems that parent Barn Swallows invested more time and energy to their chicks of highest value.
Within a breeding season, the timing of the onset of reproduction can have significant implications for breeding success. Teresa Catry (Universidade de Lisboa, Portugal) and her colleagues used a long term data set to investigate laying decisions in Lesser Kestrels. They found that individual differences in timing had substantial consequences for reproductive performance, with a significant seasonal decline.
Birds of many species attempt to defend their chicks and eggs against potential predators, and some ground-nesting birds, like the Kentish Plover, are well known for their distraction displays. Miguel Gómez-Serrano and Pascual López-López (University of Valencia, Spain) found that males that displayed greater defence behaviour were likely to mated to females that did the same, and that more defence did result in greater survival of eggs and chicks.
In a laboratory setting, Julie Gibelli and Frédérique Dubois (Université de Montréal, Canada) studied Zebra Finches, quantifying now long it took each bird to forage near a novel object in the vicinity, and challenged individuals with learning tasks. Zebra Finches that were less fearful of new things were also less flexible in their behaviour.
There are many reasons why a researcher might choose to study birds, rather than animals of other sorts. They are comparatively large, not particularly secretive, active during the day, exhibit a wide array of interesting behaviours, and are frequently quite numerous. In the field of behavioural ecology, for instance, we owe much of our understanding of important principles to our study of birds.
Photo credits: Black-throated Green Warbler, © Brian E. Small, www.audubon.org; Kentish Plover, female demonstrating the broken wing display, © Jaysukh Parekh “Suman”, www.orientalbirdimages.org
Week 38 – 02 April 2017:
The Contribution of Birds to our Understanding of the World
The world can be a frustratingly complicated place. Those who choose to go beyond an acceptance of things as they are, and attempt to determine why the world is one way and not another, will always be challenged. Among the natural sciences, physics, chemistry, geology and astronomy are each baffling in their own way, but when it comes to mind-bogglingly difficulty, biology must surely lead the way. Life adds a degree of uncertainty that can drive biologists to the brink of despair. Of all the branches of the biological sciences, the single field that is most likely to leave a biologist staring blankly at a computer screen covered in numbers is behavioural ecology.
Behavioural ecology is an attempt to understand the behaviours of animals on the basis of the evolutionary history of the group and the ecological circumstances of the individual. Why do individuals behave differently when faced with the same challenge? Why does an individual act differently when conditions change?
Many scholarly journals publish the results of research in the field of behavioural ecology. One of the most august of these is the aptly named Behavioral Ecology. It is the journal of the International Society for Behavioral Ecology, and is now in its twenty-eighth year of publication, with six issues each year. The first issue of Volume 28 arrived on my desk recently, composed of thirty-nine research articles over 345 pages.
Here is the curious part. We share the world with millions upon millions of animal species, but there are only 10,000 species of birds. Despite this, fully one-third of all the articles in that issue of Behavioral Ecology were about birds. As with so many other branches of biology, we owe much of our understanding of the world to studies of birds. For instance…
Mathieu Giraudeau (University of Zurich, Switzerland) and his colleagues reported that, at least for Japanese Quail chicks, it is important that quantities of testosterone and carotenoids deposited in the egg by the hen be balanced. Too much of either results in chicks hatching at a lower weight.
According to research by Vittorio Baglione (University of Valladolid, Spain) and his co-workers, parasitic Great Spotted Cuckoos are more likely to lay their eggs in the nests of Carrion Crows than in the nests of Eurasian Magpies in northern Spain, even though magpies were superior hosts. The unexpected result was explained in terms of how well magpie nests were concealed in the two regions.
Birds that are able to occupy both forests and cities face very different physical environments. The reproductive performance of Great Tits in forests and cities in Germany faced differences in temperature, humidity, noise and light, but Philipp Sprau (University of Munich) and his colleagues found that those differences were not sufficient to explain variation in clutch size.
In African widowbirds and bishops, the colour red is known to be important in competition among males for territories. Calum Ninnes (University of Gothenburg, Sweden) and his crew found that by increasing or decreasing the colour red in feathers beyond its natural range, they could increase or decrease the probability that a male Red Bishop would acquire a territory.
In the next entry of A Traveller’s Guide to Feathers, I will summarize additional new understandings about the behavioural ecology of birds.
Photo credits: Japanese Quail – 123RF.com; Red Bishop - © Steve Garvie, https://www.beautyofbirds.com/northernredbishops.html
Week 37 – 26 March 2017:
The Value of Jhum to Birds
To paraphrase Douglas Adams’ description of airports, it can hardly be a coincidence that no language on Earth has ever produced the expression “As pretty as an oil palm plantation.” I have walked through oil palm plantations in southeast Asia, and I have flown over them. On no scale are these plantations anything other than ugly.
And yet in world where a growing human population puts increasing demands on the landscape, monoculture production of plants such as oil palms and teak are seem as an economic saviour. Cutting down native forests and replacing them with these plantations is certain to force out creatures that are committed to live in the forests. Is there a reasonable alternative?
In the north-eastern states of India lie forests that are recognized as global biodiversity hotspots. Traditionally the indigenous tribal people of the region have utilized a system of shifting agriculture known as jhum cultivation. This form of agriculture involves a systematic rotation of land cleared of trees for cultivation, resulting in a mosaic of active and fallow fields, and regenerating and mature forests.
Jaydev Mandal of Gauhati University and Shankar Raman of the Nature Conservation Foundation in Mysore, India, set out to compare the use of mature forests, jhum, and oil palm and teak plantations by forest birds. Their study was conducted in the Dampa Tiger Reserve, the largest protected area in the state of Mizoram. It is a rugged and hilly region. The core of the reserve is free of human settlement, but a number of villages are found around the reserve’s margin. Increasingly the jhum system of cultivation in the area is being replaced by monoculture plantations, with the support of the state government.
Mandal and Raman conducted surveys in March and April of 2014, when the area was occupied by both migrant and resident birds. They recorded over 1300 individuals of 112 species, most of which were forest specialists including the Red-headed Trogon, the Gray-throated Babbler, the Wreathed Hornbill, and the Pale-chinned Blue-flycatcher.
It will surprise no one that oil palm plantations supported the fewest bird species. These plantations have almost no native trees, and are structurally simple. Teak plantations were a bit more useful to birds. Again it is no surprise that the intact native forests had the greatest number of birds of the greatest number of species. “Jhum was thus intermediate between plantations and mature rainforests in habitat structure, bird community composition, and conservation value,” wrote Mandal and Raman.
What does the future look like for birds in north-eastern India? Nothing beats an original intact operating ecosystem, but we live at a time when that is not always an option. Currently the Mizoram State Government’s New Land Use Policy aims to eliminate jhum cultivation, replacing it with other land uses. Given the apparent value of jhum for birds like the Pale-headed Woodpecker and Yellow-vented Flowerpecker, perhaps the state government might help farmers to refine their practices, rather than replace that form of cultivation.
Mandal, J., and T. R. Raman. 2016. Shifting agriculture supports more tropical bird forest birds than oil palm or teak plantations in Mizoram, northeast India. Condor 118:345-359.
Photo credits: Red-headed Trogon, photographed by/© Peter Ericsson (www.pbase.com/peterericsson/profile) – www.phuketbirdwatching.com; Yellow-vented Flowerpecker, photograph/© by wokoti - www.flickr.com/photos/wokoti/4821960569
Week 36 – 19 March 2017:
A Vulture in the Next Life
If I am to be reincarnated, I hope that I will not be brought back as a Cape Vulture. They must have great personalities, because they certainly don’t get dates on the basis of their looks. The scientific name of the Cape Vulture, Gyps coprotheres, is based on Greek words with imply that they hunt dung. They don’t hunt dung, of course, but rather scavenge carcasses of medium and large animals, which doesn’t seem like much of a step up. Even though they nest on cliffs, eggs and chicks are vulnerable to predation by eagles, crows, ravens and baboons. All in all, it sounds like a pretty tough life.
Considered to be endangered by the International Union for the Conservation of Nature, and with a rapidly declining population throughout southern Africa, a group of experts enumerated the forces working against the Cape Vulture. These included inadvertent poisoning of their food, inadequate availability of carrion during the chick-rearing phase, disturbance at their breeding colonies, habitat loss, persecution, electrocution, drowning, lead poisoning, and hunting because of a mistaken perception that eating them will have medicinal or psychological value.
Some experts see the provisioning of Cape Vultures with food at “vulture restaurants” as one of the keys to their survival. The goal is to direct birds away from uncontrolled sources of food, and provide them with predictable, poison-free nutrition. Do these restaurants serve their intended purpose? Studies of other vultures have generally reported a positive outcome. Does this also apply to the provisioning of Cape Vultures?
Dana Schabo and her colleagues recently reported on a twelve-year study of supplementary feeding at a breeding colony of Cape Vultures in KwaZulu-Natal Province in South Africa. Given that an adult pair and their single chick require about 230 kilograms of meat, and that the colony consists of forty-nine breeding pairs and about two dozen non-breeders… well, that quickly adds up to a lot of food. The vulture restaurant has been supplying Cape Vultures since 2001. On the menu are carcasses of cows, pigs and plains zebras, supplied by livestock farmers and a local conservation organization. Feedings varied, depending on the availability of carcasses.
The good news is that supplementary feeding during the nest building part of the year had a positive influence on the number of breeding adults. Schabo et al. felt that additional food permitted adults to enter the breeding season in better condition, allowing them to choose to breed, rather than waiting for conditions to improve. More food, more breeding. Curiously, providing extra food during the incubation and chick-rearing stages did not result in a higher probability of individual breeding success. It could be that not enough food was supplied to meet the needs of a colony full of growing chicks, particularly since the food supply was irregular. Alternatively, the vulture restaurant may have attracted potential predators of vulture eggs and chicks.
When I contacted her, Schabo provided me with further details of the work of the Cape Vulture team. These birds appear to play an important ecological role, but their “scavenging service is completely underappreciated and understudied.” One of the keys to the project was contact with a sugar cane farmer, Mike Neethling. Schabo described him as a passionate bird enthusiast who also happened to have a Cape Vulture breeding colony on his land. Since starting a vulture restaurant more than fifteen years ago, Neethling has made notes on every carcass set out as food, as well as breeding records for the vultures. “Mike is the hero of the story!” wrote Schabo. Because Cape Vultures cannot rip into a carcass by themselves, Neethling goes so far as to cut them open. Watching the resulting frenzy of feeding vultures must be a magnificent sight.
Local vulture enthusiast Andy Ruffle jointed the research team in 2014. He spends endless hours in a hide at the breeding colony, documenting the behaviour of these fascinating birds. Ruffle and Neethling take interested people to the breeding cliffs, detailing the ecological roll of the vultures, and explaining why it is important to protect them.
We will need additional details about the lives of Cape Vultures if we are to be sure of their long-term survival. Since 2012, the research team has been fitting individuals with satellite transmitters to track their movements. The primary focus is young birds because their mortality is so high after leaving the colony. The data is currently being analyzed, and should soon be ready for publication.
You can read more about the important work on these vultures at www.gyps-corotheres.net. Despite the supplemental feedings, I still don’t want to be reincarnated as a Cape Vulture.
Schabo, D. G., et al. 2016. Long-term data indicates that supplementary food enhances the number of breeding pairs in a Cape Vulture Gyps coprotheres colony. Bird Conservation International doi.org/10.1017/S0959270915000350.
Photo credits: Cape Volture perched on “vulture restuarant” sign – www.restafrica.org; Cape Vulture stamp from South Africa – www.pinterest.com
Week 35 – 12 March 2017:
To Feed, Or Not To Feed
Do you provide food for wild birds in your yard? If so, then you are not alone. It seems that nearly half of all householders in Great Britain feed birds, and as many as 53 million households in the United States do the same. Sunflower seeds, nyger, millet, wheat, fat, artificial nectar… it all adds up to an industry worth hundreds of millions of dollars each year. The joy that results when our feathered friends arrive in our yards to feast cannot be overstated. Birds feeders provide opportunities to develop a greater sense of empathy with wild birds. Similarly, there is little doubt that feeding birds in the non-breeding season adds substantially to their over-winter survival.
Despite all of these positive outcomes, is it possible that we are doing more harm than good by feeding wild birds during the breeding season? Could it be that bird feeders create a situation where predation of eggs and chicks increases?
Hugh Hanmer and his colleagues at the University of Reading in the UK addressed this important issue. Rather than struggling with the logistical and ethical challenges associated with predators and real bird nests, they constructed artificial nests from wire mesh, and lined them with dried grass. These were meant to mimic the nests of Eurasian Blackbirds, and were attached to trees at an appropriate height. Each nest received a pair of Japanese Quail eggs. Feeders were installed nearby, either empty, filled with peanuts, or filled with peanuts but fitted with a guard to exclude grey squirrels and large birds such as Eurasian Magpies. Each nest was monitored by a camera that was equipped with a motion-activated trigger.
Of 102 experimental nests, the contents of seventy-four were preyed upon. Those nests that were located close to filled feeders were substantially more likely to have been visited by magpies, squirrels or European Jays, although the difference between predation rates near guarded and unguarded feeders was not statistically significant.
Mark Fellowes, another member of the research team explained to me that the initial idea for the project came to him while watching birds from his kitchen window. ” I have bird feeders in my back garden, and watched a Great Spotted Woodpecker fly from a peanut feeder to a wooden nest box which had a brood of Blue Tit chicks. Within a few minutes it had managed to excavate a new entrance, and then quickly dispatched the chicks.”
“The urban landscape presents a challenging environment for wild birds to live and breed in,” wrote Hanmer et al. Urban dwelling birds may find limited natural foods, and a large number of predators including cats. Despite these challenges, urban environments do support large numbers of wild birds, and the species richness can be high.
To feed, or not to feed? Fellowes’ advice is to continue feeding birds, but to place feeders as far as possible from breeding habitat in the when birds are reproducing. He puts his feeders in front of his house in the breeding season, away from the shrubs and trees used as nesting sites in the back garden. He has also replaced all of his wooden nest-boxes with woodcrete versions to discourage predation by woodpeckers. “I get huge pleasure from feeding birds in my garden,” said Fellowes, “and I want them to benefit from my decisions; I’m pretty sure that everyone who feeds birds feels the same way.”
Hanmer, H. J., R. L. Thomas, and M. D. E. Fellowes. 2016. Provision of supplementary food for wild birds may increase the risk of local nest predation. Ibis 159:158-167.
Photo credits: squirrel-proof feeder – mailshop.co.uk; multi-level feeder – www.amazon.com
Week 34 – 05 March 2017:
One Hundred and Ten Years in the Life of a Chinese Robin
One day you might happen to find yourself travelling on the Northern Line of London’s underground subway system, approaching The Tottenham Court Road station. If someone happens to spill their cappuccino all over your shirt, you could find yourself in need of a new garment. Fear not. You will find a Primark clothing store right across the street from the entrance to the tube station.
That has not always been the case. One hundred and ten years ago, Restaurant Frascati stood where Primark is today. On the 20th of February 1907, forty members of the British Ornithologists’ Club, along with ten guests, gathered at Restaurant Frascati for the 130th meeting of the club. After finishing their dinner and completing the usual business of the club, several members brought out stuffed specimens of some particularly interesting birds. Boyd Alexander displayed six specimens including a male Black-shouldered Nightjar from Africa. Walter Rothschild introduced club members to a stuffed male Dusky-headed Parakeet from Brazil.
Ernst Hartert then brought out a stuffed adult male Rufous-headed Robin, a species that was new to science. The top of its head and the back of its neck were reddish-orange. It had a broad black stripe across its face and around its throat. The rest of the body was varying shades of black, grey and brown. Hartert explained that the specimen had been collected in the Tsin-ling Mountains of northern China, and that three specimens of the species had been deposited in the Natural History Museum’s collection at Tring.
More than a century later, the Rufous-headed Robin remains one of the least-known birds in the world. The International Union for the Conservation of Nature considers the bird to be endangered with a decreasing population. The species has not been observed at the site from which it was first described since its original collection.
When it comes to conservation, that is not sufficient information. Consequently Min Zhao of the Institute of Zoology, Chinese Academy of Sciences, and colleagues endeavored to reveal more about the lives of Rufous-headed Robins, which are found over large parts of Asia. Their findings appeared in a recent publication.
Using DNA from a blood sample collected from an adult male captured in 2015, the team determined that the bird’s closest relative is the widely-distributed Rufous-tailed Robin. Zhao et al. also studied recordings of songs from Rufous-headed Robins, and found them to be most similar to the Ryukyu Robin, a near-threated species from Japan.
In the last twenty-five years, the bird has only been observed in two spots in Sichuan Province. The team considered the habitat and environment of the eight known breeding sites of the Rufous-headed Robin, in the hope that other potential breeding sites of this cryptic species might be revealed. Their modelling suggested that additional breeding sites might be searched for in the mountains of central and northern Sichuan, southern Shaanxi, southern Gansu, and south-eastern parts of Tibet.
We have come a long way since Hartert’s first description of the Rufous-headed Robin. The work of Min Zhao and colleagues revealed new information that may assist in the bird’s long-term conservation. They warned, however, that efforts in China to help this long-distance migrant may not be sufficient. “The fate of the species may depend more on conditions and threats along its migration route and in its wintering quarters.” With only three Rufous-headed Robins ever seen outside of China, two wintering in Malaysia and one migrating through Cambodia, much work remains.
Zhao, M., P. Alström, R. Hu, C. Zhao, Y. Hao, F. Lei, and Y. Qu. 2016. Phylogenetic relationships, song and distribution of the endangered Rufous-headed Robin Larvivora ruficeps. Ibis 159:204-216.
Photo credits: photograph (c) Pete Morris, Birdquest – www.surfbirds.com; painting by John Gerrard Keulemans, from Ibis (1906) – www.wikipedia.com
Week 33 – 26 February 2017:
It’s Time!
Scott Johnson in a bird biologist at Towson University in Maryland. About ten years ago, Johnson’s wife, Bonnie, came into the house after watching a pair of House Wrens feeding their newly-hatched chicks in the backyard. Bonnie asked “If only the female incubates, then how does the male know that the eggs have hatched so that he can start bringing food to the young?” Digging through the literature, Johnson found that this brief but crucial period in the reproductive cycle had been the subject of only a single study, conducted half-a-century earlier by the eminent natural historian Alexander Skutch. More investigation was needed.
Johnson and his colleagues have been studying cavity-nesting birds such as House Wrens and Mountain Bluebirds in the central Bighorn Mountains of Wyoming for many years. Long-term investigations of that sort allow researchers to ask more and more sophisticated questions of their study species. For the purposes of these investigations, it is fortunate that wrens and bluebirds are willing to nest in artificial nest boxes. In a recent publication, Johnson et al. addressed the question of how male Mountain Bluebirds know when it is time to begin feeding their chicks.
The team investigated three possible cues for the transition in behaviour. 1. Is it possible that males notice a change in the behaviour of their mates, allowing them to know that the eggs have hatched? Females delivering food, or taking egg shells from the nest could be a good indicator of hatching. 2. Do females produce some sort of visual or acoustic signal to indicate that it is time for the male to be involved? 3. Could it be that a male will only begin to bring food to the nest when he has sensed the newly hatched young for himself? The cue might be the begging vocalizations of the four-to-six new chicks, or the sight or smell of them in the nest.
Johnson et al. used videorecorders to document activity at twenty-four Mountain Bluebird nest boxes. They found no evidence that special displays or vocalizations were given by females. Although some males saw their mates carry eggshells from the nest, or food to it, the researchers found no clear evidence that this was sufficient to cause males to bring food. In all 24 cases, males only began delivering small food items to the nest after they had had the opportunity to sense the chicks for themselves.
Johnson explained to me that one of the most striking features revealed by the research was the incredible variation in behaviour among males. “Some of our males needed but one brief encounter with the nestlings, and perhaps just the sound or smell of them, before they started hauling food back to the nest at a furious pace. Other males entered the next box repeatedly over several hours before the motivation to feed finally kicked in. There are all kinds of potentially intriguing reasons for these differences in male behavior.”
Is this situation the norm in songbirds? Future studies are needed. Additional investigation will also reveal whether a change in a male’s circulating hormones occurs when the transition to feeding begins, and if a male with greater breeding experience makes the transition more quickly.
Johnson described to me his feelings about field work on a high mountain plateau. “The wildflowers are absolutely spectacular. There is abundant wildlife. There were a number of times during the summer that we couldn’t change tapes and batteries in the camera because there was a moose standing near it. I learned a long time ago that you cannot “shoo” a moose… It makes for good stories and, at dinner parties, it certainly makes you appear vastly more interesting than you actually are.”
He is, of course, being far too modest. Field biologists are a pretty interesting group. Be sure to invite one to your next dinner party.
Johnson, L. S., C. L. Connor and A. V. Nguyen. 2016. The discovery of hatching and transition to feeding by male Mountain Bluebirds. J. Field Ornithology 87:384-390.
Photo credits: male Mountain Bluebird – www.pinterest.com; Mountain Bluebirds at a nesting box, photograph by S. and R. Proulx - www.allaboutbirds.org
Week 32 – 19 February 2017:
Small Parasites with Big Consequences
In the previous entry, I described Peregrine Falcons as big, bold and beautiful. Peregrines have been hunted in Europe in the past. The release of hydrocarbons into the environment in decades gone caused eggshell thinning and mortality for both embryos and adults. Despite this, the world population of these wonderful birds now appears to be safe. Even the capture of wild Peregrines on the Arabian Peninsula for use in falconry is having no noticeable influence on their abundance. That is not to say that the life of a Peregrine Falcon is always easy.
Rankin Inlet, a hamlet on the northwestern shore of Hudson Bay in Nunavut, Canada, has about 2500 residents. The region is also home to a dense breeding population of Peregrine Falcons which have been studied intensively since 1982. Starting in 2007, Peregrine nesting sites in the area have been monitored using motion-sensitive cameras. In most seasons, cameras capture the comings and goings of Peregrine parents and their chicks; incubating, brooding, begging, feeding, and so on. The 2013 breeding season was different, as reported by Alastair Franke, principal investigator at the Arctic Raptors Project, and his colleagues.
A pair of Peregrine were in attendance at a monitored nest of four eggs, two of which hatched on 12 July, and the other two a day later. Franke and his team visited the nest on 19 July, and the chicks appeared to be developing normally. When the nest was visited again seven days later, the chicks were gone, and the parents were not in attendance. The nestlings were far too young to have fledged. What had happened?
Images from the cameras revealed the gruesome story. On 20 July, the chicks were being fed and brooded in typical fashion. Footage from 17:15 showed that blood-feeding black flies had descended on the chicks. Within two-and-a-half hours, bleeding lesions could be seen. By 21:00, “all four nestlings displayed widespread, uniformly distributed hemorrhagic lesions over the body and multiple coalescent head lesions.” An hour later, the camera switched to infrared mode, and continued to document events at the nest. Over a thirty minutes period starting at 03:30 the female removed the bodies of all four chicks, now dead, from the nest.
Cameras at other nest sites also showed black fly attacks in 2013. Of ten nests monitored, thirteen of thirty-five chicks died. A less serious outbreak of blood-feeding flies had occurred in 2012, resulting in the death of seven nestlings. In no other year was any Peregrine chick mortality linked to black flies.
Could the deaths of young Peregrines as a result of biting flies be linked to global climate change? Average summer temperatures in the study area have risen by about 1.5 degrees C over the past three decades. Heavy rainfall events have also become more frequent. Franke et al. explained that periodic black fly outbreaks may be nothing more than normal but infrequent events resulting from a particular combination of rainfall and temperature. Either way, it seems that blood-feeding black flies are an important component of the breeding biology of birds in the Arctic.
Franke, A., V. Lamarre, and E. Hedlin. 2016. Rapid nestling mortality in Arctic Peregrine Falcons due to the biting effects of black flies. Artic 69:281-285.
Photo credits: Peregrine Falcon perched on a hotel roof in Manchester city centre - www.rspb.org.uk; nest with chicks - elasharespace2015.wikispaces.com
Week 31 – 12 February 2017:
The Wanderer
By anyone’s standards, the Peregrine Falcon is a remarkable bird. They number somewhere between 140,000 and 440,000 individuals, and occupy an area of approximately 387 million square kilometres. There are more than a dozen subspecies of Peregrine Falcon, found in nations from Afghanistan to Zimbabwe, from Moldova to Mozambique. When an individual goes into a hunting stoop, it may achieve speeds of up to 350 km, faster than any other animal. Peregrines are big, bold and beautiful. But sometimes the most interesting details are the tiniest ones.
Consider the delightfully-named Marmaduke Tunstall. A British ornithologist, in 1771 he formally described the Peregrine Falcon for the first time. As part of his description, he gave it the scientific name Falco peregrinus. The first part of the name is straightforward, based on a Latin word for sickle – a reference to its curved talons. The second part of the name is derived from another Latin word meaning stranger or wanderer. According to a dictionary of scientific bird names by James Jobling, this is a reference to a belief in the world of falconry that a young bird captured on its first migration is better suited to the sport than a bird taken from a nest as a youngster.
Falconry continues to be a part of the cultural heritage of people of the Arabian Peninsula, and Peregrine Falcons remain a key species. Traditionally, a falcon would be caught on its southward migration, held during the winter falconry season, and then released so as to avoid the trouble of keeping them through the long, hot summer, as indicated by Tunstall. In Arabia today, falconry uses both wild-caught and captive-bred birds.
A recently-published paper by Aleksandr Sokolov of the Russian Academy of Sciences and his colleagues reported on Peregrines breeding on the Yamal Peninsula of northwestern Siberia, Russia. Falcons breed at low density. Even so, Sokolov et al. documented 59 nesting attempts in 16 breeding ranges between 2008 and 2014. Some of their study involved tracking the movements of birds using satellite transmitter, but some of the work was decidedly less high-tech. Aggressive nest defense of the adult falcons made it possible to get a close look at them. The researchers found that some birds were wearing jesses as used in Arabian falconry. A jess is a short leg-strap made of silk, leather, cotton or nylon. It is used to fasten the falcon to a leash.
The sample size of birds wearing jesses in the study was understandably small, but the stories of individual birds were remarkable. In one case, a female at a nest with four chicks in 2013, and three eggs in 2014, had a blue leg band as well as jesses. The leg band had the telephone number and name of the falconer in Arabic script. When contacted, the falconer indicated that he had captured the bird as an adult on the Red Sea coast of Saudi Arabia in September 2011, but had lost it while hunting about six weeks later.
It is unlikely that the Peregrine Falcons observed in Russia had been intentionally released while still wearing jesses. Like the breeding female described, they were likely escapees. One of the most important messages to come from the research of Sokolov et al. is that these falcons have the potential to breed after a period of use in falconry. The International Union for the Conservation of Nature considers the Peregrine to be of least concern, and with a stable population. In some regions, their populations seem to be increasing. “It appears that the current harvest level of peregrines for Arabian falconry has not prevented breeding population increases and is unlikely to be currently limiting the breeding population… in its northern Eurasian breeding sites.”
Sokolov, A., V. Sokolov, and A. Dixon. 2016. Return to the wild: migratory Peregrine Falcons breeding in Arctic Eurasia following their use in Arabic falconry. J. Raptor Research 50:103-108.
Photo credits: Arab falconer with Peregrine Falcon – www.greenprophet.com; Peregrine Falcon stamp, USSR – www.shutterstock.com
Week 30 – 05 February 2017:
New Truths About Old Birds
The pages of august scholarly journals allow us to know what ornithologists were thinking about 150 years ago. Anyone interested in wading through old copies of such journals will find that bird biology then was very much like bird biology today. Our academic forbears were concerned with the lives of birds, and what caused those lives to be one way and not another. Once in a while, something completely new pops up.
In 1866, a paper appeared in Ibis, the journal of the British Ornithologists’ Union, describing a new species. The author, D. G. Elliot, wrote:
“I am indebted to the kindness of M. Jules Verreaux for the placing in my hands of a new Polyplectron, which in general appearance compares favourably with the handsomest of the known species of the genus. It was sent to the Museum of Paris from Cochin China by M. Germain, after whom I have the great pleasure of naming it.”

Hand-coloured lithograph after Joseph Wolf by J. Smit (from Elliot’s ‘A Monograph of the Phasianidae or Family of the Pheasants’)
Elliot provided the new scientific name, Polyplectron germaini, followed by a very brief description of the bird in Latin, a slightly longer English description of its appearance… and that was all. The report occupied less than a page. Elliot didn’t even reveal what sort of bird it was, assuming, I suppose, that everyone was familiar with the genus Polyplectron. He didn’t have anything to say about the bird’s natural history or abundance. I think the story is worth expanding upon.
The accepted common name for the new bird is Germain’s Peacock-pheasant. Daniel Giraud Elliot (1835 – 1915) was an American zoologist with professional ties to Paris and London. The gentleman who showed Elliot the stuffed bird was Jules Pierre Verreaux (1807 – 1873), a professional collector of natural history artifacts. By 1866 Verreaux had risen to the post of assistant curator of the museum that housed the pheasant, the Muséum national d’Histoire naturelle. The specimen remains in the museum, catalogued as number 1865-577.
The specimen of Germain’s Peacock-pheasant appears to have been collected in Cochinchina, representing the southern third of what is now Vietnam. In 1866 it was a French colony where Louis Rodolphe Germain (1827-1917) served as a veterinary surgeon to France’s colonial army. He probably shot and stuffed birds in his spare time.
This peacock-pheasant is a slightly chunky ground-dweller in Vietnamese forests. Overall it is black and brown, but with numerous large iridescent spots on its back, wings and tail. These spots appear green in some light and blue in others. The beautiful tail is between 22 and 35 cm (9 and 14 inches) long. Females are smaller and a bit duller than males. Its diet is a mystery, and everything we know about its breeding habits comes from birds in captivity.
As far as I can tell, only one additional scholarly paper has ever been published about Germain’s Peacock-pheasant. Destiny Omeire and his colleagues at Texas Southern University and the Houston Museum of Natural Science collected a sample of tissue from a museum specimen. When I asked about the origin of the specimen, Dan Brooks at the HMNS explained that it came from captive stock, rather than from the wild. Omeire and crew extracted genetic material from sub-cellular structures known as mitochondria, determined the complete sequence of the DNA’s constituent parts, and reported their findings in the journal Mitochondrial DNA. The paper’s corresponding author, Hector Miranda, wrote to say that students in his laboratory are sequencing DNA in other peacock-pheasants, and that he is preparing a manuscript “about the phytogeography and pattern of evolution of Polyplectron of Southeast Asia, as driven by climate change since the Pleistocene.”
One hundred and fifty year on, ornithologists are uncovering wonderful new things about birds discovered long ago.
Elliot, D. G. 1866. On a new species of the genus Polyplectron. Ibis 8:56.
Photo credits: Hand-coloured lithograph after Joseph Wolf by J. Smit from Elliot’s ‘A Monograph of the Phasianidae or Family of the Pheasants’ - www.donaldheald.com; Germain’s Peacock Pheasant by Louis Agassiz Fuertes – www.wikimedia.org
Week 29 – 29 January 2017:
I Wonder if Mousebirds Hate Cat Stevens?
Providing for the needs of zoo animals involves much more than a suitable diet, adequate space, and proper veterinary care. Captives require an environment that is enriching. This is particularly true of birds and mammals that may develop behavioural problems in an impoverished environment. Stereotypic acts and self-harm are among the possible outcomes if individuals are not stimulated sufficiently, and in suitable ways.
As a consequence, zoos engage in programs of behavioural enrichment. To date most of these programs have been directed at mammals, particularly primates and carnivores that have particularly complex social behaviours in the wild. Less attention has been paid to the value of enrichment efforts for birds, even though birds and mammals often share the same zoo enclosures.
Recently the Buffalo Zoological Gardens in Buffalo New York studied the value of acoustic enrichment on their captive gorillas. Lindsay Robbins and Susan Margulis of Canisius College in Buffalo took the opportunity to study the responses to enrichment of three species of birds that shared the gorilla exhibit. These were a pair of Lady Ross’s Turacos, two Spectacled Mousebirds, and a Superb Starling. All of these birds are native to Africa.
Over a period of weeks, behaviours of the birds in silent control periods were compared to their behaviours during three acoustic experiences. The first was playback of a compact disk called “Sounds of the African Rainforest.” The second experience involved an assortment of compositions by the classical composer Frédéric Chopin. Finally the birds (and gorillas) were observed during playback of music by the British rock band Muse. That band was chosen because their music contained “no vulgar language or loud, cacophonous sounds.”
Robbins and Margulis documented that nature sounds caused an increase in the amount of flying by members of all three bird species. In terms of flying, the Starling seemed to like Chopin, but none of the birds was keen on Muse. Rock music caused both the starling and mousebirds to vocalize less, but resulted in a significant increase in the performance of vocal duets by the turacos. The researchers concluded that acoustic enrichment of this sort may have a positive benefit on birds held as captives in zoos, without adding substantially to the work load of keepers.
Muses’ 2015 album, Drones, won a Grammy Award for Best Rock Album. They will be headlining at music festivals in Leeds and Reading later this year.
Robbins, L., and S. W. Margulis. 2016. Music for the birds: effects of auditory enrichment on captive bird species. Zoo Biology 35:29-34.
Photo credits: Spectacled Mousebird, photograph © Mike Haworth – howieswildlifeimages.com/2014/11/26/mousebirds/; Lady Ross’s Turacos - www.zoochat.com
Week 28 – 22 January 2017:
From England, to the Sinai, and Beyond
For birds of many species, migration is not optional. In the non-breeding season, conditions on their breeding territories become so extreme that either their needs are not met or their tolerances are exceeded. An English country garden, alive with birdsong in spring is likely to fall silent when winter drives birds south in search of more clement circumstances. Fly away, little feathered friend, or suffer the consequences.
The biggest challenge of migration may not be the journey itself, but the need to find suitable stop-over habitat along the way. If the migratory route is a long one, birds will need to stop for rest and replenishment along the way. Suitable habitat for a short break can be hard to find, particularly if much of the migratory route is over desert.
Andrew Power, Olivia Norfolk and Francis Gilbert of the University of Nottingham in England recently published a paper concerning the value of Bedouin gardens in the southern Sinai to both migratory and resident birds. Power and crew were not in Egypt to just to study birds; the main purpose of their expedition was to collect data on a rare butterfly, the Sinai hairstreak, whose populations are considered to be vulnerable to extinction by the International Union for the Conservation of Nature. Since the researchers were in the region anyway, they took advantage of the opportunity to record their sightings of birds.
The work was completed in the Sinai’s St. Katherine protectorate, essentially a national park, but with added importance. Some birds were noted in the irrigated, managed gardens of the local Bedouin people. Other birds were seen in valley beds and on mountain slopes, habitat which is not managed for agriculture.
In response to an inquiring email message, Power described the circumstances of his field experiences. “I spent long periods of time camping with the Bedouin in the mountains, or in a tourist camp run by the Bedouin.” The camp was very close to St. Catherine’s Monastery, which Power visited several times.
Over a five month period, the research crew documented fifty-one species of birds. Of these, 65% were migrants. Birdlife in the managed gardens was particularly rich. Among the frugivorous birds drawn to the apricots, plums and mulberries of the garden were Golden Orioles and Hooded Wheatears. Garden insects drew in Spotted Flycatchers and Common Redstarts. The gardens were important to resident species too; Collared Doves, Laughing Doves and House Sparrows were seen there, but never in unmanaged habitat. Beyond food and water in the gardens, some bird species utilized Bedouin structures. Power explained to me that Rock Martins and White-crowned Black Wheatears constructed their nests in the walls and on the eaves and sides of houses, taking advantage of almost any crevice.
The St. Katherine protectorate is clearly important for birdlife in the area, and Bedouin gardens, described by Power et al. as “oasis-like,” seem particularly important throughout the year. The Bedouin people collect rainwater to increase the productivity of their mountain gardens, and this action provides much-needed resources for both resident and migratory birds.
A surprisingly large proportion of the migratory bird species that were seen in the Sinai are known to bird enthusiasts in England. If global climate change causes Egypt to become hotter and drier as predicted, the gardens managed by Bedouins may become even more important for wildlife. From the bird’s point of view, it is regrettable that traditional gardening practices are becoming more difficult as Bedouins come to rely more on paid employment. Water is a limited commodity, and tourism and an increasing local population are likely to put further demands on that precious resource.
Power, A., O. Norfolk and F. Gilbert. 2016. The oasis effect: bedouin gardens benefit resident and migratory birds in southern Sinai, Egypt. Sandgrouse 38:7-11.
Photo credits: Dr Andrew Power, https://twitter.com/aerpower?lang=en (set of three photos above) and https://crowcragproductions.com/photos-2/egypt/ (Caspian Tern, below)
Week 27 – 15 January 2017:
Doing the Best With What We Have
Every Sichuan Partridge in China; every Tepui Tinamou in Venezuela; every Chaplin’s Barbet in Zambia… each is ruled by a tiny voice at the back of its head. It is the voice of natural selection, driving it forward. The voice instructs all living creatures to make the maximum possible genetic contribution to following generations. “Reproduce! Reproduce!” cries the tiny voice.
The last 75 years of research has shown that the voice of natural selection is incredibly nuanced. The drive to pass along our genetic legacy is not simply a matter of producing as many offspring as possible, as often as possible. For instance, there is no point in a pair of White-crowned Sparrows producing eight chicks if they can only feed five. A female Welcome Swallow should not produce twelve eggs if all are so small as to be inviable. If a pair of Atlantic Puffins successfully raised three chicks instead of one, but the parents died from exhaustion at the end of the breeding season, the tiny voice would likely be displeased.
In their attempt to pass along their genes, all creatures are faced with tradeoffs. Some of these tradeoffs were illustrated neatly in a recently-published study of Yellow-billed Gulls in northern Africa by Addessalem Hammouda and his colleagues. They observed the reproductive efforts of the gulls at a colony on the Mediterranean coast of Tunisia. At 131 nests, Hammouda et al. noted the days on which each egg was laid, the volume of each egg, and its fate.
The majority of Yellow-billed Gulls at this colony had a three-egg clutch, with a lesser number producing two eggs. The volume of these eggs varied considerably from 61 to 107 ml (2.1 to 3.6 ounces). Hammounda and his team found that the last-laid egg in the clutch was likely to be the smallest. They also discovered that the volume of eggs in three-egg clutches tended to be larger than those in two-egg clutches. Over the 27-day incubation period, 92% of all eggs hatched successfully, but that success was higher among three-egg clutches. What does it all mean?
In the Yellow-billed Gull, last-laid eggs tend to produce the last-hatched chicks. When food is scarce, the slightly-younger, slightly-smaller chicks tends to be outcompeted by their siblings. It seems as though females are investing more resources in their first eggs because those are likely to produce chicks with a higher probability of surviving.
Creating eggs is almost certainly demanding for female birds. It seems that the female gulls that are sufficiently robust to lay three eggs are also in sufficiently good condition to create larger eggs. Females who are able to create three large eggs are also better able to defend those eggs against predation than females producing two smaller eggs. Curiously, terrestrial predators such as snakes and rodents were very rarely seen in the vicinity of the gull colony. However, predation of eggs by other Yellow-billed Gulls was seen frequently.
Although the birds in the Tunisian study were not marked for individual identification, and so the ages of individuals were not known, it is likely that age plays a role in reproductive tradeoffs. Older, more experienced birds are likely better able to acquire resources and defend their nests than younger birds. Beyond that, an individual that is approaching the end of its life may hear the little voice say: “What are you waiting for? This could be your last chance!” Hammouda et al. explained that older females are “likely to have lower residual reproductive potential,” and so should “invest more heavily in current breeding attempts.”
The little voice is subtle, but it is also relentless.
Hammouda, A., F. Hamza, J. Pearce-Duvet and S. Selmi. 2016. Relationship between clutch size, egg volume and hatching success in a Yellow-billed Gull Larus michahellis colony in south-eastern Tunisia. Ostrich 87:139-144.
Photo credits: Yellow-billed Gull stamp - birds-on-stamps.com; photograph of Yellow-billed Gulls by Cosmin-Ovidiu Manci - birds.nature4stock.com
Week 26 – 08 January 2017:
A Celebration of Good News
In 1908, a writer named Minori Ogawa published an article in the scholarly journal Annotationes Zoologicae Japonenses. The paper described birds from the southern islands of Japan. It seems that a Mr. Alan Owston of Yokohama had employed two men to shoot birds to add to his burgeoning collection of stuffed specimens. Between May and December of 1904 the collectors gathered specimens of 124 bird species.
According to Ogawa’s article, the collection included a thrush previously unknown to science. Ogawa described the Amami Thrush as being similar to White’s Thrush, but with twelve tail feathers instead of fourteen, and with a thicker, longer and darker bill. Ten specimens of this thrush were added to Owston’s collection. The bird was, and still is, known only from a single island, Amami-Oshima.
Leap forward a century…
The Amami Thrush, now considered to be a subspecies of the Eurasian Scaly Thrush, was doing very poorly. In 1996 the estimate of its abundance was just 58 individuals. Found on just a single island, the habitat favored by the Amami Thrush, old-growth broad-leafed forests, had been subject to clear-cut harvest between the late 1950s and 1970s. Far fewer trees meant far fewer thrushes.
And if that weren’t bad enough, thirty small Indian mongoose had been released on Amami-Oshima in 1979 in hopes that they would control populations of a venomous snake. In twenty years, the mongoose population grew from thirty to over 6,000 individuals. If you were a hungry mongoose, would you rather tackle a deadly venomous pit viper or a harmless little thrush?
Now comes the good news, courtesy of a recently published paper by Taku Mizuta and his colleagues. As a result of an eradication program that began in the year 2000, the population of mongooses has been significantly reduced. When contacted, Mizuta explained to me that it was likely that the last mongooses would be eliminated from Amami-Oshima by 2023. The large-scale clearance of mature forests on the island has declined since the 1970s, and it appears that habitat suitable for Amami Thrushes may be recovering. Have the birds responded to the change in circumstance?
Among the difficulties of assessing the abundance of these forest-dwelling thrushes is their timidity. It is simply a hard bird to spot. Fortunately, male Amami Thrushes have a loud, distinctive and melodious voice. Dedicated members of the Amami Ornithologists’ Club have, year after year, been engaged in formal censuses of their thrush. Starting before sunrise, they have been walking transect lines, listening for the thrush’s song, and plotting their observations on maps. In other areas, club members stood in particular spots for 50 minutes, documenting all of the Amami Thrushes heard.
Mizuta and his colleagues described the results of these surveys. They worked from the reasonable assumption that only adult male Amami Thrushes sing, and that females and immature males do not. They concluded that the thrush population has now climbed to about 5,000 males and females capable of breeding. Formally thought to be critically-endangered, as a result of the efforts of members of the Amami Ornithologist’s Club we can now say that the population is at least stable or may still be increasing.
Mizuta et al. wrote: “We intend to continue this public-participation survey for many years as a means of promoting further conservation of the Amami Thrush.” Has awareness of the plight of the bird increased? Mizuta wrote to me to say that the local newspaper and radio station have been reporting the results of the surveys each year, allowing locals to better identify with the birds. The social situation also seems to be changing for the better. The Japanese government is applying to have the Amami and Ryukyu islands added to UNESCOs list of World Heritage sites, and locals are showing more interest in the natural history of their part of the world.
All of that seems to be worth celebrating.
Mizuta, T., et al. 2016. Song-count surveys and population estimates reveal the recovery of the endangered Amami Thrush Zoothera dauma major, which is endemic to Amami-Oshima Island in south-western Japan.
Photo credits: Scaly thrush - zipcodezoo.com/index.php/Zoothera_dauma; Scaly thrush stamp – colnect.net
Week 25 – 01 January 2017:
Seabirds Improve the Lives of… What Did You Say?
Forgive yourself if you have never heard of tardigrades; most people haven’t. Also known by the common names “water bears” and “moss piglets,” chances are very high that you are currently sitting just a few metres from members of one or more species of tardigrade. Even so, the probability that you will ever see a tardigrade is vanishingly small.
Tardigrades themselves are vanishingly small; most species are invisible with anything less than a very high quality microscope. You can’t eat them, and they can’t eat you. They don’t spread diseases, nor do they have any marketable qualities. Researchers have described about 1200 species of tardigrades, and it is entirely possible that you will never hear the word “tardigrade” ever again. But that doesn’t mean that they aren’t interesting to bird enthusiasts.
Little Auks (Alle alle) are, as their name implies, small seabirds. They eat plankton, and breed colonially in the high Arctic. They are among the most abundant seabirds in the world, and so despite their diminutive size, Little Auks have the potential to greatly modify the local environment around their colonies. As with other breeding seabirds, some of the nutrients they extract from the sea are defecated over land, changing the chemistry of the soil.
Spitsbergen Island is tucked neatly between the Arctic Ocean and the Greenland Sea, and is administered by Norway. A Polish research station has been operating at Hornsund, a fjord on southern Spitsbergen, for nearly sixty years. In 2016, Krzysztof Zawierucha of Adam Mickiewicz University in Poznań and his colleagues reported on a study of the impact of breeding Little Auks on the tardigrade community of Hornsund.
Zawierucha and his team collected moss and lichen along two transects, each a kilometre-long. One of these transects stretched from a colony of 30,000 Little Auks down to the sea. A second transect, not influenced by the Little Auk colony, acted as a control. Could it be that the enrichment of the soil downhill from the seabird colony resulted in more tardigrades? Might the species composition of the two sites be different?
Peering through their microscopes, Zawierucha and company found 1990 tardigrades from 32 different taxonomic groups with enchanting names like Isohypsibius elegans and Microhypsibius bertolaniii. Tardigrades were more abundant in the vicinity of the Little Auk colony than in the region not influenced by the seabirds, making this report the first to find a positive effect of seabird faeces on tardigrades in a polar region. Zawierucha et al. concluded that fertilization of the soil by the seabirds enhanced plant growth, which created small-scale climates that were more favorable for tardigrades.
Representatives of fifteen tardigrade taxonomic groups were found in both situations, but ten were found only along the control transect, and seven were found only along the transect leading from the Little Auk colony. Some of the differences might be attributable to small-scale differences in the environment, such as greater acidity of the soil downhill from the seabird colony. Some of the tardigrade species collected in this study had never been recorded in the region before, illustrating our lack of knowledge about many aspects of polar biology.
When I contacted Zawierucha, he explained that his main research interest was tardigrades, concerning himself with their ecology, taxonomy and biogeography, but that he was very fond of other invertebrate groups as well. He is interested in glaciers and creatures that live on glaciers as models for life that may exist on other planets. “When I saw glaciers for the first time,” he wrote, “I realised that they define rules of biodiversity and isolation.” Whether it is seabirds or miniscule animals that few people have heard of, field biologists have no end of exciting research ahead of them.
Zawierucha, K., K. Zmudczyńska-Skaarbek, Ł. Kaczmarek, and K. Wojczulanis-Jabukas. 2016. The influence of a seabird colony on the abundance and species composition of water bears (Tardigrada) in Horsund (Spitsbergen, Artic). Polar Biology 39:713-723.
Photo credits: light micrograph of a tardigrade, photograph © Sinclair Stammers – www.extremetech.com/extreme/218492-tardigrades-already-impossible-to-kill-also-have-foreign-dna; Little Auk - www.newhorizonsonline.co.uk/gallery-items/little-auk/
Week 24 – 25 December 2016:
Things We Know and Things We Don’t
Birds are, without a doubt, the best known creatures in the world. Most are brightly-coloured, reasonably large and abundant, display interesting behaviours, and are particularly active during the day. Consequently birdwatching is a hobby; wormwatching isn’t. Birds have been under scientific scrutiny for centuries, and much of what we know about the natural history of animals in general is based on what we first learned about birds.
Some bird species are much better studied than others of course. Thousands of scholarly publications have considered the lives of Mallards and Barn Swallows, partly because they are both common species found in places with lots of people to watch them. At the other end are Black-browed Babblers and Cayenne Nightjars about which we know almost nothing, particularly because they are extraordinarily rare. They reside in countries with very few bird biologists, and may have already fallen to extinction.
In terms of our knowledge of natural history, most species of birds fall somewhere in the middle. They are reasonably common and we know quite a bit about them. There are, however, exceptions.
The International Union for the Conservation of Nature considers the Stripe-cheeked Woodpecker, Piculus callopterus, to be of least conservation concern. A species endemic to Panama, they are not wildly common and have a patchy distribution, but at least their numbers seem stable. So why is it that we know so little about them? In part the answer is that the forests of the foothills of eastern Panama are very difficult to access.
Until recently no one had ever reported seeing the nest of one of these Panamanian woodpeckers. A 2015 publication by William Adsett and Leslie Lieurance changed all of that. In April and May of 2013, Adsett and Lieurance had the great fortune to observe the comings and goings of a pair of Stripe-cheeked Woodpeckers attempting to raise chicks in forest in Chagres National Park in the foothills of eastern Panama.
By great fortune, the pair of birds choose to excavate a nesting cavity in a Cecropia tree very close to Adsett’s home. When I contacted him, Adsett explained that he and his wife own a property within the Altos de Cerro Azul, a residential development of 2000 lots within Chagres National Park. This region represents one of the highest and wettest parts of the entire watershed of the Panama Canal. “We are fortunate in having the Stripe-cheeked Woodpecker frequently in our bit of the forest,” Adsett explained, “but it was sheer luck that the pair nested in sight of our house.”
A combination of direct observation and time-lapse video footage provided Adsett and Lieurance with our first insights into the private lives of breeding Stripe-cheeked Woodpeckers. For instance, the opening to the nesting cavity was 3.4 metres (11 feet) from the ground where the tree was 20 centimeters (8 inches) in diameter. It took nine or ten days from the start of excavation until the first egg was laid, and incubation lasted 12 or 13 days. Both parents incubated the eggs and fed the young, but it was the male that made the greatest number of feeding trips and spent each night in the nesting cavity.
I wish that I could report a happy ending, but I can’t. The young woodpeckers died 17 or 18 days after hatching, which was about a week before they might have been expected to leave the nest. Although they could not be certain, Adsett and Lieurance felt that failure was likely attributable to predation. A large column of army ants was observed to enter the nest, but whether they were responsible for the death of the chicks is unknown. It is possible that a nocturnal predator took the nestling, either dead or alive, after the ant attack.
“It is our guess that this species do not generally return to the same nest hole for a second or third attempt – the trunks just deteriorate too fast.” Adsett explained that cavity-nesting in the tropics can be a precarious business. The excavation of a different nest in a dead tree by the same species caused the trunk to snap off just days after completing the hole.
Adsett, W. J. and L. Lieurance. 2015. First breeding record, vocalisations and morphology of Stripe-backed Woodpecker Piculus callopterus, a Panamanian endemic. Cotinga 37:79-86.
Photo credits: Stripe-cheeked Woodpecker, © Paul Jones – www.flickr.com/photos/paulbjones/24765456272; drawing of a Stripe-cheeked Woodpecker - www.hbw.com/species/stripe-cheeked-woodpecker-piculus-callopterus
Week 23 – 18 December 2016:
The Lives of Birds are Rarely Simple
In eighteen century Europe, when songbirds disappeared from their breeding grounds in autumn, it was hard for many people to believe that they flew thousands of kilometres to spend the winter in more clement regions. Some eminent naturalists imagined that small birds such as swallows burrowed into the mud at the bottoms of rivers and lakes in autumn, only to emerge unharmed in the spring.
Even after centuries of study, many aspects of the migratory behaviour of birds remain a mystery. It is that mystery that makes migration a rich field of study, open to both high-tech and low-tech approaches.
A recent publication by Andrea Harnos and her colleagues at Szent István University, the Hungarian Natural History Museum, and Eötvös Loránd University, all in Budapest, revealed the wealth of information that can result from a low-tech approach to the study of migration. Their report described the result of a study of the small European Pied Flycatcher between 1989 and 2014 as the birds migrated between breeding grounds in Europe and wintering areas in Africa. For 26 years, volunteers erected nets, captured flycatchers, measured them, and documented their age and sex before sending them on their way. The study was conducted in Danube-Ipoly National Park in central Hungary.
Over a period of 26 years, data was collected for 2323 Pied Flycatchers. Among the discoveries of Harnos et al. was that males on their spring migration passed through the study area 11 days earlier in 2014 than they had in 1989, but that females had not changed the timing of their northward migration. No significant changes occurred in the timing of autumnal migration by either sex over the course of the study.
The wings of spring migrants averaged 2 mm longer at the end of the study, and earlier migrants had significantly longer wings than birds who passed through the study area later in the season. In contrast, the wing length of autumn migrants did not change over the study, and earlier arrivals had shorter wings. Of spring migrants, 60% were male, but females accounted for 61% of autumn migrants.
At first glance, this might seem like a confusing collection of results, but Harnos and her colleagues believe that they have untangled the story. In terms of timing, males that arrive on the breeding grounds earlier gain an advantage over later males in terms of attracting a mate; climate warming has likely allowed their average migration dates to shift to earlier dates. For some reason females did not complete their northward migration earlier, so neither breeding nor southward migration could happen earlier.
Other findings are also likely to be related to climate change. Pied Flycatchers have a vast breeding distribution in Europe and Asia, and populations differ slightly in body size. Changes to climate have likely caused shifts in the migration routes of many bird species, such that the research group in Hungary were probably capturing Pied Flycatchers from different populations at the beginning and end of the study. Differences in sex ratio between spring and autumn probably reflects the complex nature of migration, with male and female flycatchers taking different routes between breeding and overwintering grounds.
Anyone seeking simple answers should probably avoid the field of avian migratory behaviour. Even so, you can rest assured that songbirds do not spend the winter underwater.
Harnos, A., Z. Lang, P. Fehérvári and T. Csörgő. 2015. Sex and age dependent migration phenology of the Pied Flycatcher in a stopover site in the Carpathian Basin. Ornis Hungarica 23:10-19.
Photo credits: male Pied Flycatcher – drawing of male Pied Flycatcher - www.rspb.org.uk/birds-and-wildlife/bird-and-wildlife-guides/bird-a-z/p/piedflycatcher/; photo by Rob Vaasse, The Netherlands, May 2003, www.birdforum.net/opus/European_Pied_Flycatcher
Week 22 – 11 December 2016:
Silly Bird, Smart Bird
Hornbills are ridiculous. Just look at them. They are among the silliest-looking creatures ever. Outrageous they may be, but they are not dimwitted when it comes to solving the challenges of life.
There is a fundamental principle in community ecology that states that perfect competitors cannot coexist. If members of two species attempt to exploit the same essential, limited resource in the same place at the same time in the same way, one will be eliminated. This ecological principle is so entrenched in the discipline that it even has a name – competitive exclusion.
How then do three species of large hornbills, all doing essentially the same thing with their lives, manage to co-exist in the tropical forests of far-northeastern India? Rohit Naniwadekar of Manipal University, Charudutt Mishra and Aparajita Datta of the Nature Conservation Foundation in Karnataka set out to discover how these hornbills exploit limited food resources in slightly different ways, allowing them to occupy the same regions at the same time. This was part of Naniwadekar’s Ph.D. study.
The three bird species under consideration were the Great Hornbill (considered by the IUCN to be near-threatened), Wreathed Hornbill (of least conservation concern), and the Rufous-necked Hornbill (vulnerable to extinction). The study was conducted over a period of more than two years in the Namdapha Tiger Reserve in Arunachal Pradesh. In what ways did these birds differ in the way that they exploited fruit in forest trees?
After tramping endless kilometres along forest trails, Namiwadekar and his coworkers found subtle differences in the diet and behaviours of the hornbills that allowed them to defeat competitive exclusion. Although all three species had a varied fruit diet, Great Hornbills spent much more time dining on figs than they did on non-fig fruits. Upon finding a suitable fig tree, they spent an average of 21 minutes feeding there before moving on. Wreathed and Rufous-necked hornbills spent a greater proportion of their time foraging from non-fig fruit trees, where they averaged 43 minutes and 10 minutes respectively.
Wreathed and Rufous-necked hornbills behaved differently in their search for food, allowing them to both exploit non-fig fruit trees. Wreathed Hornbills travelled long distances in search of fruit, which was energetically costly but provided abundant opportunity to find areas with the highest fruit availability. In contrast, Rufous-necked Hornbills were more sedentary, and kept themselves well informed about the availability of fruit over a much smaller home range. Even though they had similar diets, Wreathed and Rufous-necked Hornbills employed different solutions to the trade-off between travel costs and foraging efficiency.
Given that these species have somewhat different diets, and exploit food resources in different ways, Naniwadekar et al. concluded that: “the three sympatric hornbills play a complementary role in seed dispersal.”
When I contacted her about the hornbill study, Datta explained that it is necessary to follow the birds for long periods in order to understand some aspects of their behaviour; this is particularly difficult in dense tropical forests. Hornbills “are wary of people due to hunting in most areas… There are often physical and logistical challenges working in remote tropical forests, where research is mostly on foot, heavy rains often disrupt work, malaria is common, while leeches and ticks… are also common.
Of the hornbills the group studied, the Rufous-necked is in the greatest peril. They have been extirpated from Nepal, and have a very restricted distribution in India. They may be found at altitudes as high as 2000 metres. Most regrettably, many of the forests at higher elevations are not legally protected, and the Rufous-necked Hornbill is hunted by some of the tribal communities for their beaks and feathers. Studies like the one by Naniwadekar and his colleagues will provide insights into hornbill biology that could have implications for their conservation.
Naniwadekar, R., C. Mishra and A. Datta. 2015. Fruit resource tracking by hornbill species at multiple scales in a tropical forest in India. Journal of Tropical Ecology 31:477-490.
Photo credits: Rufous-necked and Wreathed Hornbills - orientalbirdimages.org (Rufous-necked Hornbill (c) Ateeb Hussain; Wreathed Hornbill (c) Wong Tsu Shi); Great Hornbill – Pinterest
Week 21 – 04 December 2016:
A Bird Too Beautiful
Physics, chemistry and astronomy are simple. Each of these disciplines is governed by a limited number of physical laws. If the chemical naphthalene melts at 80.2 degrees Celsius today, it will melt at 80.2 degrees Celsius tomorrow. If Mars takes 24 hours, 37 minutes and 22 seconds to rotate on its axis this week, the same will be true next week.
Biology is difficult. At every turn, life introduces uncertainty, meaning that if 1435 Pied Wagtails show up at a nocturnal roost in Scotland tonight, we cannot know with absolute certainty how many will show up tomorrow night. When biology become conservation biology, the world becomes a ridiculously complicated place. Every conservation issue involves a tangle of interactions among countless species, interactions between those species and their non-living world, and complications of economics, ethics and politics.
Surely there is no place in the world where conservation biology is more complex than China. Considering birds alone, the nation has almost 1400 species, seventy-five of which breed in China and nowhere else. Among China’s endemic birds, the Sichuan Partridge, Hainan Peacock-pheasant, Silver Oriole and Blue-crowned Laughingthrush are all endangered and declining in number. The Manchurian Reed-warbler, Grey-hooded Parrotbill, Cabot’s Tragopan and many more are considered vulnerable.
Chunfa Zhou and Zhengwang Zhang of Beijing Normal University and Jiliang Xu of Beijing Forest University recognized the need for up-to-date information on the abundance and distribution of Reeves’s Pheasant. Endemic to central China, this pheasant was, comparatively recently, abundant and distributed widely. As a result of illegal hunting and habitat destruction and fragmentation, Reeves’s Pheasant is currently recognized to be Vulnerable by the IUCN. Zhou and her colleagues asked if that category was appropriate.
In the search for the pheasant, field surveys were supplemented by interviews of local residents, elders, village heads and hunters, and with personnel in county forestry bureaus. Sightings of birds along transect lines, the sound of the pheasant’s “wing-whirring displays,” and the presence of moulted feathers were all used as evidence of ongoing occupation of an area.
The results of Zhou et al. were not encouraging. Since earlier surveys, 46% of sites had lost their Reeves’s Pheasants, and many other populations had declined in abundance. The human population continues to grow in the area, and economic development plans are likely to make the situation worse in the future.
Zhou et al. wrote: “The long tail feathers of the male Reeves’s pheasant are widely used for decoration of Chinese opera costumes… and consequently a great number of Reeves’s pheasants are hunted.” These birds may be too attractive for their own good. Poaching was apparent at 83% of surveyed sites, and poisoning at 20% of sites. “Farmers in China deliberately poison Galliformes (grouse, pheasants etc.) to prevent them from eating crop seeds,” wrote Zhou et al. In rural areas, it seemed that people were not conversant with national policies on wildlife, and conservation education programs directed at small towns did not get the message to those in remote rural situations. The booming Chinese economy has led to ongoing deforestation of the exactly the sort of habitat preferred by Reeves’s Pheasant.
As the Chinese government works to upgrade its conservation policies, it is important that the best estimates of abundance and distribution be considered. Zhou and her colleagues suggested that the status of Reeves’s Pheasant be upgraded to a first-grade nationally protected species, and that the IUCN change the bird’s status from vulnerable to endangered.
Zhou, C., J. Xu and Z. Zhang. 2015. Dramatic decline of the vulnerable Reeves’s Pheasant Syrmaticus reevesii, endemic to central China.
Photo credits: Reeve’s Pheasant - www.tumblr.com; Reeve’s Pheasant stamp - sandaproject.blogspot.com.au
Week 20 – 27 November 2016:
Then and Now
Many fields of scientific endeavour have changed dramatically in my lifetime. New technologies have allowed great advances in astronomy, physics, chemistry, and some branches of biology. Ornithology less so. I think the reason is that biologists studying birds acquired most of the tools that they needed early on. Binoculars, a notebook and pencil, a rain jacket and hat… What else do you need?
It is true that our capacity to track individual birds has increased greatly as a result of advances in transmitter technology, but many studies continue to use nothing more sophisticated than numbered aluminum leg bands. Progress in gene sequencing has allowed us to leap forward in our understanding of evolutionary relationships among birds groups, but ornithologists still return to museum collections to measure stuffed specimens.
If we go further back in the history of ornithology, differences between then-and-now become more apparent. Papers concerning bird biology published a century ago were far less numerical than they are today, and statistical analysis of data was rare. It also seems to me that research then was less likely to be directed at answering fundamental questions.
For more than 130 years, The Auk has been one of the three great journals dedicated to research on birds. In the January 1916 issue was an article by Henry J. Fry concerning seasonal declines in birdsong. The study was conducted at the field station at Cold Spring Harbor, Long Island, New York. Although Fry was not absolutely clear, it seems that he was teaching a course in ornithology at Cold Spring, and had his students collect observations. The songs of birds were noted each day between 06:00 and 07:30, and between 10:00 and 11:30, in addition to alternating afternoons. The study ran from July 1 to 10 August.
One by one, Fry described the lessening of singing by birds as the season progressed. “The Scarlet Tanager (Piranga erythromelas)” wrote Fry, “was in full rich song till about July 16, though further study may show this day to be inaccurate by several days. From that time on the song declined steadily and the last one was recorded on the twenty-seventh.”
“The Wood Pewee (Myiochanes virens) began its decline almost imperceptibly around July 20, and from that day it gradually became less and less, though the daily diminution was scarcely evident.” And that was that. Fry did not speculate about the cause of seasonal variation in the volume and frequency of singing, nor about possible reasons for species’ differences in behaviour. It wasn’t science. At best it was natural history.
But consider this… It appears that global climate change may be causing some birds to alter their behaviour, such that they breed earlier in the year. In 1914 Henry Fry generated a systematic data set of the singing behaviour of more than two dozen species of birds. In doing so, he created an opportunity. A dedicated student of birds could reproduce Fry’s study to look for changes more than 100 years later. The Cold Spring Harbor Laboratory is still there to facilitate study.
My efforts to find out more about the life of Henry Fry have resulted in little success. He may have been at Columbia University in some capacity in the 1890s. He appears to have been an associate member of both the American Ornithologists’ Union and the Delaware Valley Ornithological Club at the time he wrote his paper on singing behaviour. He may eventually be commemorated by a student keen to replicate his work. Are there any takers?
Fry, H. J. 1916. A study of the seasonal decline of bird song. Auk 33:28-40.
Photo credits: Scarlet Tanager stamp - colnect.com; Wood Pewee – www.birdfreak.com
Week 19 – 20 November 2016:
When Bigger is Better
The differences in nest architecture among bird species are remarkable. At one end are the nests of Killdeer, constituting no more than a few bits of gravel scraped into a circle around a slight depression in the ground. At the other end are the gigantic nests of some raptors that grow in size with the passing years as the resident pair adds more and more material.
As with so many things in life, nests represent a tradeoff for the birds constructing them. More time and effort spent on building the perfect nest might result in better survival of the eggs and chicks, but this could come at the expense of time not spent in self-maintenance and foraging. Biologists are interested in how individuals resolve such tradeoffs. What is the optimal solution? Which alternative will result in the greatest lifetime reproductive success?
Michał Glądalski and his colleagues at University of Łódź in Poland are involved in a long-term study of birds utilizing artificial wooden boxes for nesting. Since 2012, Glądalski and his crew have been studying Great and Blue tits nesting in boxes in two habitats in central Poland. A mature deciduous forest contains 300 nest boxes, while a parkland site with fragmented tree cover has 200 boxes. To these nest boxes, tits add moss and dry grass to complete an outer structural component, and hair and feathers as an inner lining. As measures of reproductive success, the number of eggs in the nest, the proportion of eggs that hatched, and the proportion of hatchlings that eventually fledged from the nest were all documented.
Compared to most songbirds, tits lay a lot of eggs. The average clutch size of Blue Tits varied between ten and thirteen; Great Tit clutch sizes averaged between eight and eleven. Nests constructed within boxes by Blue Tits were considerably larger and heavier than those of Great Tits. Nests did not differ significantly between the two habitat types.
The key finding of the study was that, in general, the greater the mass of the nest lining, the greater the reproductive performance of the parents. Overall nest size and mass was also important. In tits, the nest serves to help maintain an optimal warm and humid environment for the eggs and young nestlings. Where nests are concerned, bigger appears to be better.
Or so it would seem. Studies to date have not fully disentangled the relationship. Nests characteristics alone may not explain everything. It is possible that parents that are more experienced or in better physical condition are able to construct better nests, but are also better able to care for their eggs and chicks. Is nest size the crucial factor, or is it the effort made by the parents? “Future experimental studies… should at least try to efficiently separate these two factors,” wrote Glądalski et al.
It is possible for a researcher to become so focused one particular aspect of their topic that the “bigger picture” is lost. Glądalski and his team are considering the lives of birds utilizing nest boxes in Poland from many different perspectives. When I contacted him, Glądalski explained that he and members of his group are considering breeding success, physiological stress, the influences of heavy metals, variation in egg size, and the genetics of individuals in their population. Glądalski is particularly interested in the ways that global climate change might influence the timing of reproduction and nest characteristics. I suspect that this sort of comprehensive approach is likely to result in a more profound understanding of bird biology in the long term.
Glądalski, M., et al. 2016. Effects of nest characteristics on reproductive performance in Blue Tits Cyanistes caeruleus and Great Tits Parus major. Avian Biology Research 9:37-43.
Photo credits: Blue tit coaxing a chick to fledge, photo by Andrew Williams - www.bbc.co.uk/nature/21445671; Great tit and chicks - www.audubon.org/news/build-nest-box-welcome-spring-birds
Week 18 – 13 November 2016:
Did I Just Hear An Explosion?
In an environmental sense, a catastrophe is a sudden and violent change, unpredictable and experienced infrequently, but resulting in widespread damage. Earthquakes, cyclones, tsunamis, cataclysmic floods and wildfires create such rapid changes in the physical environment as to challenge the ability of plants and animals to respond. Should I stay, or should I flee?
With the eruption of the Puyehue volcano in northwestern Patagonia on June 4 2011, Pablo Alarcón of the National University of Comahue in Argentina and his colleagues were handed a unique opportunity to study the responses of Andean Condors. By chance, ten adult condors had been fitted with GPS tags seven months before the eruption, allowing the researchers to follow their movements with precision. Following the volcanic eruption, ten more condors were given tags.
When the volcano erupted, it ejected 950 million tons of ash into the atmosphere, with an initial column reaching 12 km (7.5 miles) into the sky. Drifting to the east, the ash blanketed 24 million hectares (92,000 square miles). Silica was the major constituent of the ejected ash, which caused blindness, gastric distress and death to livestock, particularly sheep, over the following three months.
Inhabitants of the Andes Mountains, Andean Condors are large, mobile scavengers that feed mainly on carcasses of large and medium-sized mammals. Like sheep. So when the Puyehue volcano erupted, condors in the area faced a choice. Should they stay in the area and take advantage of a sudden rich feeding opportunity, but risk being harmed by the ash, or should they leave behind the habitat that they knew well in an attempt to live out the catastrophe elsewhere?
Alarcón and his coworkers predicted that condors would change their behaviours over time, initially avoiding the potential damage of the volcanic ash, later moving back to take advantage of the opportunity to eat dead livestock, but then avoiding those affected areas after the food had been depleted.
Did that happen? “Interestingly,” wrote Alarcón et al., “no strong eruption-induced effects were apparent on the movement behavior of the condors studied… Overall, our results suggest that the studied birds maintained their general movements patterns after the volcanic eruption and remained largely exposed to the conditions imposed by the catastrophe.”
Perhaps conditions were not sufficiently severe after the eruption to change condor behaviour. Maybe the availability of food is not limiting for condors in this region, so that they didn’t need to take advantage of additional dead livestock. It could be that the advantages of being in an area and social situation that they knew well were greater than the risks of exposure to ash. Abandoning a breeding area comes with the risk that the individual might never get it back.
For the livestock of the region, the eruption of the Puyehue volcano was a catastrophe. For Alarcón and his colleagues, the eruption was serendipitous.
Alarcón, P. A. R., et al. 2016. Movement decisions in natural catastrophes: how a flying scavenger deals with a volcanic eruption. Behavioral Ecology 27:75-82.
Photo credits: Andean Condor stamp from Argentina – www.stampboards.com; Puyehue volcano eruption – www.guardian.co.uk
Week 17 – 30 October 2016:
Where Did You Say It Is?
There cannot be too many bits of land that humans have never visited. There are, however, places that people have found and occupied, but later abandoned and forgotten. In contrast, birds tend to discover sites, settle in, and stay. Some of us dream of travelling to sites that have been abandoned by everything except birds.
Swains Island is just such a locale. A circular atoll in the South Pacific Ocean, the island is part of the Tokelau group, which are a dependency of New Zealand. Swains Island itself is administered by the United States under the umbrella of American Samoa. It is, quite frankly, hard for me to imagine why the United States would want anything to do with Swains. The island is about 2 km long and 1.5 km wide, with a central lagoon full of brackish water. Nowhere does the island poke more than 9 m above the sea, and drinking water is hard to collect. The island’s dominant plant is the coconut palm, but shorefront shrubland, marsh and forest can also be found. Although Swains Island was a coconut plantation in the past, and was occupied year-round, it is now completely uninhabited.
Except, of course, for periodic visits by biologists curious to see which creatures call the island home. The most recent visit was by members of the Department of Marine and Wildlife Resources in American Samoa in September of 2012. Andrew Titmus, conducting research in American Samoa for a doctoral degree at the University of Hawaii, was invited to inventory birds on the island while other researchers looked at insects, coconut crabs, sea life and vegetation. Another key member of the group was Nicole Arcilla, the chief wildlife biologist of DMWR. The group visited Swains Island for six days, 36 years after the previous survey.
Titmus explained to me that the team got to Swains on a chartered boat that normally ferries people American Samoa and Western Samoa. “The journey took the better part of a day and once there we needed to use a small boat to ferry equipment and supplies to the island through a small opening in the reef.”
Titmus and his crew walked the coastline of the island twice, counting every bird they encountered. They documented the birdlife along nine transects through the island’s vegetated habitat, starting at the coast and ending at the central lagoon. They also spent three evenings at sea, watching for rare or transient bird species.
The efforts of Titmus et al. revealed 19 species of birds, some rare and some common. Almost all were seabirds or shorebirds. The Bristle-thighed Curlew, Sanderling, and Brown Booby were represented by just two individual each. In contrast, 7,092 Black Noddys and 4,412 White Terns were counted.
To anyone familiar with waterbirds, it is interesting that: “All nesting and roosting seabirds were observed within trees, with no birds present on the ground or beach.” Titmus and his crew attributed this behaviour to introduced predatory mammals. Even though the island’s feral pig population has been eradicated, there is still a small group of feral cats, and Pacific rats were common everywhere.
This lead Titmus et al. to a wonderful suggestion. If a concentrated effort was to eliminate the cats and rats on Swains Island, and if coconut palms were replaced by native trees that better suited birds, the island could become an important site for nesting seabirds. It is even possible that shearwaters that burrow into the soil to nest might take up residency on Swains.
Titmus wrote to say that “while we were on the island, we camped on the beach by the old village site and copra shed, and there is still the remains of a trail between the village site and the family residence site where there is a dilapidated house and a small graveyard.” To me that sounds like a pretty idyllic spot to study birds.
Titmus, A. J., N. Arcilla and C. A. Lepczyk. 2016. Assessment of the birds of Swains Island, American Samoa. Wilson Journal of Ornithology 128:163-168.
Photocredits: Map of Swains Island - www.janeresture.com/tokelau_islands/swains.htm; Black Noddy with chick by Pete Leary (US Fish and Wildlife Service) - www.usgs.gov/media/images/black-noddy-chick

Week 16 – 23 October 2016:
Irony in the World of Birds
It would probably be quicker to list all of the places where House Sparrows cannot be found. Native to Europe and large swathes of Asia, the Middle East and Africa, it has been successfully introduced to North and South America, the Caribbean, Australia, New Zealand, Hawaii, Mauritius and so on. Adaptable in the extreme, according to a 1993 book by Clement, Harris and Davis, “especially remarkable being the birds that lived and bred 640 m down a coal-mine in South Yorkshire, England, living on food provided by miners.” The closely-related Tree Sparrow also has a wide breeding distribution in Europe, Asia and the Middle East, and has been introduced to Sardinia, the United States, India, Indonesia, Hong Kong, Borneo, Australia, and, well, you get the picture.
Are you ready for some irony? Although there may be as many as 540 million House Sparrows in the world, and perhaps 570 million Tree Sparrows, significant population declines in some regions have caused concern. The British Trust for Ornithology estimates that the U.K. House Sparrow population has declined by half in recent decades, and the city of Edinburgh is said to have lost 90% of those birds between 1982 and 1998. What is going on?
Martin Šálek of the Academy of Sciences of the Czech Republic, and Jan Riegert and Stanislav Grill of the University of South Bohemia investigated House and Tree sparrows in the community of České Budějovice. With 94,000 inhabitants, it is the largest city in the Czech Republic’s South Bohemia region. Better known for its industry than its sparrows, České Budějovice is the home of Koh-i-Noor Hardtmuth pencils and the Budweiser Budvar brewery.
In May of 2013, Šálek and his crew documented every House and Tree sparrow and every nest within the 19.2 km2 (7.4 miles2) spread of the city. On foot and on bicycle, they travelled the city’s streets and parks, peeped over fences, and listened for diagnostic chirping. Dividing the city into 675 plots, each 200 by 200 m (10 acres), they noted the type of habitat found in each plot, including residential sites, parks and cemeteries, and industrial and commercial areas.
Šálek et al. recorded 2251 House Sparrows and 537 Tree Sparrows. House Sparrow densities were highest in residential areas, and were comparatively abundant in the city centre. “High” is a relative term in this case, and House Sparrow densities were much lower than in many other European cities. Tree Sparrows are less dependant on urban landscapes, preferring gardens to residential sites in České Budějovice, they were not seen in the city centre. It seemed “that both sparrows are associated with diverse and mixed habitat composition, where they can find suitable foraging and nesting conditions.” This is particularly true in the breeding season, when adults need to find insects to provide a protein-rich diet for their nestlings.
Even though they are quite flexible, both sparrows built the majority of their nests in crevices and holes in buildings. The older the building, the better. The modernization of old buildings is likely to result in population declines in these sparrows.
“Nesting opportunities could,” explained Šálek et al., “be improved by simple and cheap solutions.” In the reconstruction of old buildings, architects might allow for access for sparrow to nesting sites. New buildings could incorporate cavities for sparrows and other urban birds. Solutions to environmental problems are not always costly or complicated.
Šálek, M., J. Riegert and S. Grill. House Sparrows Passer domesticus and Tree Sparrows Passer montanus: fine-scale distribution, population densities, and habitat selection in a Central European city. Acta Ornithologica 50:221-232.
Photocredits: Eurasian Tree Sparrow stamp - philatelicbirds-art.blogspot.com.au; male House Sparrows - www.theguardian.com
Week 15 – 16 October 2016:
To Feed Or Not To Feed
In some parts of the world, people are able to add substantially to the lives of birds by maintaining birdfeeders. For a modest outlay for lumber, nails and seed we can contribute to the nutrition of granivorous birds in our yards. For a similarly-small sum spent on glassware and sugar, we might attract nectar-feeding birds. The British Trust for Ornithology is involved with bird feeding through its Garden Bird Feeding Survey (www.bto.org/volunteer-surveys/gbfs), as is the Cornell Lab of Ornithology in the U.S. through its Project FeederWatch (feederwatch.org).
Feeding birds is not a one-way gift. Feathered friends reward our generosity by living their lives a little bit closer to ours. Moving from the individual to the wider community, supporting wild birds helps to establish and maintain important links between citizens and nature.
That is not to say that feeding birds is necessarily always positive. Amoung the possible risks are increased disease transmission among birds crowding around feeders, added attention by predators, and the creation of dependency on our generosity. Could it be that feeders also result in increased bird mortality through collisions with the windows of residential buildings?
Justine Kummer, an M.Sc. student at the University of Alberta, and her supervisor Erin Bayne, investigated the possibility that a birdfeeder on a residential property could increase the likelihood of fatal collisions of birds with windows. Their study was conducted in 2014 and 2015 at 43 homes in and around the city of Edmonton in central Alberta, Canada. Some studies of bird biology are more interesting to the public than others, and the study of Kummer and Bayne attracted considerable media attention. The Canadian Broadcasting Corporation was particularly helpful in recruiting homeowners in the Edmonton area to the study. Birdfeeders were erected and stocked with seed, and collisions at windows close to feeders were compared to incidents at windows with no feeders. Homeowners were asked to make daily checks for evidence of collisions by birds.
Over the year-long study, 145 birds collided with windows. Of these, 89 survived and the outcome for another 45 was unknown; only 11 fatalities were reported. Seedeaters like House Sparrows, Blue Jays and Dark-eyed Juncos were among the birds involved in collisions. Curiously, birds not expected to eat grain sometimes struck windows, including Cedar Waxwings and American Robins. This was, presumably, not a consequence of birdfeeders.
The presence of birdfeeders roughly doubled the number of collisions with windows. However the story isn’t as simple as that. Variability in the data was considerable. Far fewer birds struck windows in winter than in any other season, even though non-migrants made greater use of feeders in winter. Of the 43 homes in the study, owners at 18 did not report even a single bird collision, but four homeowners reported a combined 76 strikes, and a single window was responsible for more than 11 collisions. “This suggests that the effects of a bird feeder are dependent on the house and the window,” wrote Kummer and Bayne. “Overall, some houses have a much higher risk than others, suggesting that impacts of feeding birds may be context dependent.”
Having successfully defended her M.Sc. thesis, Kummer is now considering the future. She wrote to say that “I’m very interested in science outreach and education, and science communication – which was a large part of my overall project.” Please join me in wishing her great success in that field.
Kummer, J. A. and E. M. Bayne. 2015. Bird feeders and their effects on bird-window collisions at residential houses. Avian Conservation and Ecology 10(2):6.
Photo credits: Blue Jay at a feeder – www.uniquebirdhouseboutique.com; Cedar Waxwing – www.allaboutbirds.org
Week 14 – 09 October 2016:
When a Tree is Not Just a Tree
Birds have an uncanny ability to fill our lives with joy by simply being there. In the depths of a Australian summer, under the cruel, baking sun of the outback, it takes just a small waterhole to attract Zebra Finches. On a dimly-lit morning in the coldest part of a Canadian prairie winter, a bird feeder stocked with seeds will attract House Sparrows. Wherever you are in the world, birds are there to be seen.
But we need to take care. The mere presence of birds does not guarantee that all is well with the world. In our near-limitless capacity to transform the landscape, we may be making subtle changes. It sometimes takes a keen eye to notice that change.
Fabrico Cid and Enrique Caviedes-Vidal of the Universidad Nacional de San Luis in Argentina recently published a paper concerning human-created changes to the birdlife of a region around a reservoir. The Embalse La Florida was constructed between 1945 and 1953, and serves as the main source of drinking water to the cities of San Luis (170,000 residents) and Villa Mercedes (111,000 residents). The reservoir has an area of more than 650 ha (1,600 acres) and a circumference of 36 km (22 miles), and provides recreational opportunities in the form of swimming, fishing, kayaking and windsurfing. That all sounds pretty good, right?
A portion of the land around the reservoir is a combination of native woodlands, Acacia shrubs and grasslands, but the remainder has been modified as small-scale tree plantations in support of campsites and a settlement. The plantations are dominated by trees not native to Argentina such as pine and eucalyptus.
Cid and Caviedes-Vidal surveyed the bird populations of the natural habitat and tree plantations in both the breeding and non-breeding seasons. They documented almost 3000 birds of 75 species. The smallest was the 5 g (0.2 ounce) Yellow-billed Tit-tyrant, and the largest was the 1.9 kg (4.2 pounds) Black Vulture. Spot-winged Pigeons were far more abundant than any other species.
In both the breeding and non-breeding seasons, bird abundance was higher in tree plantations than among native plants; species richness was similar between the two habitat types. Even though the distances between sites was short, most bird species were seen in one habitat type or the other, but not both. Cattle Tyrants, Monk Parakeets and Brown Cacholotes were only seen in the plantations, whereas Pale-breasted Spinetails and Southern Bearded-tyrannulets were seen only in the natural habitats. Plantations favored large-bodied generalist feeders; native vegetation favored smaller-bodied birds that feed within the foliage of the plants.
Changing the landscape changes the composition of the bird population within it. Although no birds of special conservation concern were recorded by Cid and Caviedes, the authors pointed out that the area covered by tree plantations could grow substantially in the near future. Loss of natural habitat is likely to have consequences for birds that rely on those conditions. With an expanding human population, it may become necessary to set aside patches of native vegetation in the region. Biological diversity is a matter of variation as well as abundance.
Cid, F. D. and E. Caviedes-Vidal. 2014. Differences in bird assemblages between native natural habitats and small-scale tree plantations in the semiarid midwest of Argentina. Wilson J. Ornithol. 126:673-685.
Photo credits: Yellow-billed Tit-tyrant © Jorge Martín Spinuzza (www.avespampa.com.ar & neotropical.birds.cornell.edu); Brown Cacholote © Karl-Fredrik Sjolund - buenosairesbirding.com/birds
Week 13 – 02 October 2016:
I Love Birds, But…
A child who wanted to create a picture of a Blue Swallow would only need one crayon – Midnight Blue. These birds are deep metallic colour all over, and sport a pair of very long tail feathers. Blue Swallows breed in southern Africa and spend the non-breeding season further north in Kenya and Uganda. Regrettably, very few children are likely to know what a Blue Swallow looks like. Just 3000 members of this species remain, and those are thinly distributed. There are more people living in Thermopolis, Wyoming, than there are Blue Swallows in all the world. I have been to Thermopolis. It isn’t big. Southern Africa is.
According to preliminary observations, it seemed that a substantial breeding population of Blue Swallows might reside in the southern Tanzanian highlands. Steven Evans and colleagues at the University of Venda, North-west University (both in South Africa), and the Tanzanian Bird Atlas Project recognized the pressing need to quantify the swallow’s abundance in the region. Ideally they would also be able to determine the bird’s habitat preferences, and current threats to that habitat.
Evans and his team conducted two surveys of Tanzania’s Kipengere Mountain Range; once in 2008/2009, and again in 2012. The team surveyed 3,635 km (2260 miles) of grasslands, pastures, croplands, villages, and tree and tea plantations. (For perspective, this is greater than the distance from Thermopolis, Wyoming, to Montreal, Quebec, or between Strasbourg in France and St. Petersburg in Russia.) For each swallow sighting, GPS coordinates, altitude and habitat type were recorded. The group found nests of Blue Swallows by searching the undersides of bridges and by asking locals.
The total of 151 Blue Swallows and 14 nests were recorded at 62 localities. Considering the total area searched, and by estimating the total area suitable for the species in that part of Tanzania, Evans and crew felt that the region was likely to support between 602 and 1362 individuals. Clearly, the southern Tanzanian highland grasslands support a significant proportion of the world’s population of Blue Swallows, particularly in natural pastures and around villages. “This finding indicates that the development of rural villages is not a threat to the birds,” wrote Evans et al. Indeed, Blue Swallows may be feeding on insects attracted to livestock and dung, and buildings in villages may provide a suitable surface for the construction of swallow nests. It is even possible that artificial structures could be erected to facilitate swallow nesting.
Evans and his coworkers described the need for similar swallow surveys in the Marungu Highlands and Upemba National Park in the DRC. If they are currently planning such work, then they must be braver than I. On its website: “Global Affairs Canada advises against non-essential travel to the Democratic Republic of Congo… points of entry from Burundi, Rwanda and Uganda should be avoided at all times because of continuing insecurity and lawlessness in those areas… Crime is endemic in the DRC as a result of extreme poverty and the prevailing climate of impunity.”
I love birds, but not enough to get killed while conducting a survey.
Evans, S. W., E. M. Baker, N. E. Baker and D. Cillers. 2016. Current distribution and population size of the Blue Swallow Hirundo atrocaerulea in the southern Tanzanian highlands. Ostrich 87:37-46.
Photo credits: male Blue Swallow, The Handbook of the Birds of the World Alive - http://www.hbw.com/species/montane-blue-swallow-hirundo-atrocaerulea; female Blue Swallow photograph by Warwick Tarboton (copyright Warwick Tarboton) – www.warwicktarboton.co.za/index.html
Week 12 – 25 September 2016:
That Seems Like a Lot of Foolin’ Around
As I described in an earlier entry, birds are a model of monogamy. There are lots of exceptions, but a bird family usually consists of a mom, a dad and a nest full of happy chicks. This system of mating is attributed to the value of having two parents attending to the needs of their dependent nestlings.
With the advent of genetic technologies, it became possible to determine with certainty whether the social father of nestlings was also their genetic father. It came as a surprise to many researchers to find that, for a wide range of bird species, chicks were frequently the product of a male other than their social father.
The benefit to males of extra-pair copulations is obvious. By sneaking on to the next territory to copulate with a female to which he is not mated gives him the opportunity to pass along more of his genes without the additional cost of caring for the young. The possible risk to the male is that while he is being sneaky with the neighbouring female, another male might be doing the same thing with his mate.
Benefits of extra-pair copulations to females are a little more subtle. By engaging in such behaviour, a female might be protecting herself against the possible infertility of her social mate. She may also be able to copulate with a male that is genetically superior to her mate, resulting in better offspring.
Members of some bird groups seem to do a great deal of foolin’ around. Studies of raptors, on the other hand, have shown that very few chicks are not genetically-related to their social fathers. In part this may be because mated birds of this sort copulate with impressive frequency. Or at least that was the story until now…
In a recent publication, Robert Rosenfield and his colleagues at the University of Wisconsin and the Alaska Science Center showed that Cooper’s Hawks, Accipiter cooperii, are an exception. Studying a breeding population of hawks in Milwaukee, USA, the team collected blood samples from as many adults and chicks as they could. They used a decoy Great Horned Owl to cause adult hawks to fly into a net so that blood samples could be taken; Rosenfield told me that the decoy’s name was LE, pronounced “Lee.” It was a small male, and so looked a bit like a Long-eared Owl.
The team found genetic evidence of a remarkably high incidence of infidelity. Nineteen percent of Cooper’s Hawk chicks in this region were not the product of their social father, and 34% of all broods contained at least one illegitimate chick. Could this be because mated Cooper’s Hawks copulate less frequently than other raptors, allowing greater opportunity for fertilization by other males? Apparently not. In an earlier publication, Rosenfield reported that in the thirty days before egg laying, pairs of Cooper’s Hawks averaged more than twelve copulations a day.
What is the cause of all this foolin’ around in this socially monogamous bird? It seems that male Cooper’s Hawks provide food to their mates, and that food delivery is often followed by copulation. Could it be that females are accepting copulations from additional males, and possibly even soliciting them, in exchange for food before the energetically-demanding period of egg production? The breeding population of Cooper’s Hawks in Milwaukee is particularly dense, and the urban environment is rich in food. Their diet includes sparrows, robins, starlings, doves and chipmunks. Rosenfield et al. suggested that this may make it easy for “females to encounter extra-pair males with prey and trade copulations for food.”
Rosenfield, R. N., S. A. Sonsthagen, W. E. Stout and S. L. Talbot. 2015. High frequency of extra-pair copulations in an urban population of Cooper’s Hawks. Journal of Field Ornithology 86:144-152.
Photo credits: Cooper’s Hawk photograph by Johann Schumacher - www.audubon.org/field-guide/bird/coopers-hawk; Cooper’s Hawk stamp – www.birdtheme.org
Week 11 – 18 September 2016:
“Over Here! Look at Me! Over Here!”
Some birds are much better-known than others. Let me offer you the following evidence. If a biologist is keen to find scholarly publications addressing a particular topic or species, she might use an academic search engine called Web of Science. Using “Turdus merula,” the scientific name of the Eurasian Robin, as a search term will return 680 recent papers, and “Zonotrichia leucophrys,” the White-crowned Sparrow, will reveal 730 publications. Neither of those come close to “Anas platyrhynchos,” the Mallard duck, which will lead to 2,649 papers.
Pity, then, the poor Pygmy Wren-babbler. When I entered its scientific name, “Pnoepyga pusilla,” into Web of Science, I was offered just two publications. One was a 2013 paper which considered the relatedness of all members of the genus. The other was a 1991 publication about an entirely different type of bird.
It isn’t that the Pygmy Wren-babbler isn’t interesting. It is. At just 8 cm (3 inches) in length, and with no tail to speak of, this wren-babbler looks very much like an olive-brown egg adorned only by a tiny black beak and oversized legs. Seven subspecies are recognized, and some of these should probably considered as full species. Four distinctive forms are found on the islands of Indonesia alone. Despite being reasonably common in Nepal and China, the little bird just hasn’t been very well studied.
In 2016, Muhammad Nazri Janra of Andalas University in Indonesia and Richard Noske of Charles Darwin University in Australia published a brief note in the journal of the Indonesian Ornithologists’ Union. In November of 2009 Janra and his field crew were using mist-nets to capture birds 1,200 metres up the side of Mount Singgalang in the province of West Sumatra. The study targeted the Sumatran Cochoa, a beautiful and vulnerable songbird known only from Sumatra. While working along a woodland trail, Janra inadvertently flushed a Sumatran Pygmy Wren-babbler (Pnoepyga pusilla lepida) off its nest. The bird hopped along the ground, dragging one wing as though it was broken; it was an act of attempted deception. Janra was able to find the nest, even though it was very well concealed. Situated 50 cm (20 inches) above the ground, and constructed of plant fibres and covered with moss, it contained two white eggs.
The broken-wing display has been described most frequently from ground-dwelling shorebirds such as killdeer and other plovers. This strategy is surely designed to draw predators away from nests, although it requires great attention by the adult if it is to avoid being pounced upon. Not only was this the first observation of the broken-wing display by any member of the genus Pnoepyga, but it represents only the second published record of breeding by the species on Sumatra. It is a reasonably common bird on the world’s sixth-largest island, and yet we know almost nothing about it.
Their work required Janra and his team to investigate other locales. When they revisited the site where the broken-wing display had been witnessed a month earlier, they were unable to locate the nest nor spot any Pygmy Wren-babblers, and so it is not clear whether the nesting attempt had been successful.
It is through the dedication of researchers like Janra that we come to a better understanding of the world of birds. One observation at a time.
Janra, M. N. and R. Noske. 2015. Sumatran Pygmy Wren-babbler Pnoepyga pusilla lepida performing broken-wing distraction display near nest. Kukila 18:62-64.
Photocredits: Pygmy Wren-babbler – drawing of a Sumatran Pygmy Wren-babbler - http://www.hbw.com/species/pygmy-wren-babbler-pnoepyga-pusilla; http://www.birdingintaiwan.com/pygmywren-babbler.htm
Week 10 – 11 September 2016:
Waste Not, Want Not
The efforts of dedicated biologists enlarge our understanding of bird biology with their discoveries. After years of work in the field and laboratory, results are published in the pages of scholarly journals so that they can illuminate others, now and into the future. That is one of the interesting things about scientific journals – with any luck, their contents will continue to be available to researchers for centuries to come.
I recently pulled a ninety-year old copy of the journal The Condor: A Magazine of Western Ornithology off a shelf in my personal library. The copy was Issue 1 of Volume 28. Along with journals like Ibis and The Auk, it represented a repository of the best new findings of bird enthusiasts, with particular emphasis on the west coast of North America.
By today’s standards, much of the content of the 56-page copy in my hand would be considered natural history, rather than science. The first thirty pages were a consideration of bird husbandry in British aviaries, written by Casey Wood of Kandy, Ceylon (now Sri Lanka). This was followed by short pieces on the appearance of Franklin Gulls in Colorado, the consumption of frozen insects by American Dippers, and the vocal behaviour of Band-tailed Pigeons. One article was particularly revealing, not just of the birds being studied, but also of the writer.
Ernest D. Clabaugh explained that he had been engaged in banding (“ringing” in the UK) nestling Western Screech-owls. He noted the remains of meals brought by the parents to their chicks. Most of these were House Sparrows, supplemented by gophers, mice and salamanders. “It is interesting to note,” wrote Clabaugh, “that practically all of the birds found in the nest had their heads eaten off when I found them.”
Even more interesting to me is Clabaugh’s honesty. At 05:30 on 3 June, 1924, he dropped one of the young owls while banding it. The bird appeared to be injured, but he applied band number 223576, and put it back in the nest. When Clabaugh returned to the nest the next day: “the leg of no. 223576, which held the band, was all that was left of this bird. It was in the nest, the bird apparently having been eaten by the three remaining young owls.”
I found a few details of the life of Clabaugh. A resident of Berkeley, he trained as a civil engineer at the University of California, but his great joy appeared to be the lives of birds. Over a period of 30 years, Clabaugh banded many thousands of birds, an activity he continued to his death in 1953. He served on the executive of the Western Bird Banding Association, and the Cooper Ornithological Club, the group that published The Condor. He was also a very honest man.
Clabaugh, E. D. 1926. Notes of the food of the California Screech Owl. Condor 28:43-44.
Photo credits: Western Screech Owl, Oregon Zoo – www.oregonzoo.org; Western Screech Owl stamp – www.pinterest.com
Week 9 – 04 September 2016:
ZOOM!
According to its website, Adolfo Suárez Madrid-Barajas Airport in Spain is: “One of the airport capitals of the 21st century.” How exciting! Its four runways have a combined length of 15.5 km (9.6 miles). Four terminals provide for 71 airlines serving 182 domestic and international destinations. Forty-six million passengers used the airport in 2015, which is more than 126,000 travellers each day. All those busy people, and a few birds.
In recent years, the impact on wildlife of noise created by human activities such as traffic and construction has become a topic of considerable interest. Birds in particular seem to be vulnerable to disturbance by human-created noise, if only because many birds use acoustic cues to help avoid predation. What better situation to study the impact of noise on birds than the nosiest spot of all – a busy airport?
In a recent publication, Ignacio Klett-Mingo and Diego Gil of the Universidad Politécnica de Madrid, and Ignaco Pavón of the Museo Nacional de Ciencias Naturales described the results of their study of aircraft noise at Madrid-Barajas airport on the local birdlife. The airport is bordered by a river and a woodland, home to a substantial bird community. When arriving and departing airplanes pass the woodland, peak noise levels can exceed 85 dB. This level of noise is apparently sufficient to cause permanent hearing damage in humans after just eight hours of exposure.
Klett-Mingo and his colleagues placed six bird feeders in the study area, stocking them with peanuts. They documented the behaviour of Great Tits using video recorders placed close to the feeders. Birds living next to a busy airport runway are unlikely to be surprised when an airplane arrives or departs, but do these events disrupt their lives?
Klett-Mingo et al. found that tits changed their behaviour significantly as airplanes passed by. Birds spent less time feeding and more time with their heads up, scanning their environment, spending almost twice as much time being vigilant when aircraft noise was greatest. It seemed as though tits found it necessary to be more vigilant when their world got noisy.
This isn’t surprising. Great Tits, like many other birds, rely on acoustical signals to help warn them about potential predators. For instance, some birds produce alarm calls in threating situations, allowing other birds around them to respond in an appropriate way. When lots of individuals are watching for danger, these signals can allow each of them to spend less time being vigilant, and more time foraging. When things get too noisy, the value of these social signals are lost, and each individual needs to be more wary.
The lost foraging time might be particularly harmful in winter when days are shorter and food is less available. Beyond food challenges, Klett-Mingo speculated that repeated loud noises might increase physiological stress in birds living near runways. Perhaps the increased vigilance by individual tits cannot fully compensate for aircraft noise, resulting in an increased risk of predation, particularly if the noise leads to distraction.
Diego Gil explained to me that there are many bird predators in the study area, including Eurasian Sparrowhawks. The main cause of this abundance is likely the absence of hunting pressure around the airport, but it is also possible that airplane noise makes potential victims more vulnerable, and so the habitat is more valuable for foraging by predators.
Birds do not wear noise-cancelling headphones. Wouldn’t it be interesting to know whether Great Tits exposed to the roar of aircraft at Madrid-Barajas Airport suffer hearing loss?
Klett-Mingo, J. I., I. Pavón and D. Gil. 2016. Great Tits, Parus major, increase vigilance time and reduce feeding efforts during peaks of aircraft noise. Animal Behaviour 115:29-34.
Photo credits: Great tits playing with a camera, photographed by Vadim Trunov – www.express.co.uk/news/nature/624939/amazing-picture-squirrel-and-bird-messing-around-with-camera; first day cover of Great tit and Hortensia stamps from Spain, sent to collector Florian Tirk – worldwidecovers.wordpress.com/2009/09/
Week 8 – 28 August 2016:
Well, That’s Okay If You Are A Seal, But…
Several years ago New Scientist magazine described Bouvet Island in the far south Atlantic Ocean as the only place on Earth not to have had any plant or animal species introduced by humans. It is also the locale for the 2004 horror/sci-fi film Alien vs. Predator. Further, it is one of the few places I can think of without a single bird that isn’t a seabird. Bouvet Island is almost entirely covered by ice and is, quite possibly, the most remote bit of land on the planet.
That did not deter Corne Niemandt of the University of Pretoria and seven of his colleagues from Norway and South Africa. They recently published a report on their findings concerning penguins that breed on Bouvet Island, also known as Bouvetøya. The team visited the largest ice-free spot on the island during four penguin breeding seasons between 1997 and 2008. They were particularly interested in the food brought to Chinstrap Penguin and Macaroni Penguin chicks. If we hope to understand the possible impacts of global climate change on wildlife species, then we must first understand fundamental features of their biology such as what they eat.
Adult penguins and their chicks were counted, and samples were collected from the stomachs of adult penguins during the chick-rearing period. Niemandt et al. found that Chinstrap Penguins were extremely picky eaters. Of 56 samples, all contained a type of krill with the scientific name Euphausia superba, and this one creature constituted more than 99% of the mass of stomach contents. The study population consisted of between 350 and 500 Chinstrap Penguins from 1996 to 2001. When counted again in 2008, the combined population of adults and chicks had declined by more than 80%.
Macaroni Penguins at Bouvet were more flexible in their diet. Krill were still a common food, but a range of fish species such as Electrona carlsbergi and Krefftichthys anderssoni were also eaten. The population of Macaroni Penguins declined, but not as severely as did Chinstraps.
What caused the decline in Penguin populations? Perhaps food was became less abundant, either because of physical changes in the environment or increased competition with other species such as krill-feeding whales. Niemandt and his colleagues found that most penguins were able to maintain two chicks each year, “suggesting that breeding birds are finding enough food.” Instead, it seems likely that declines in breeding penguin numbers resulted from a lack of breeding space. The breeding population of Antarctic fur seals has increased in recent years, and their colony now occupies almost all of the available space, displacing the penguins. Bouvet Island has the world’s second largest Antarctic fur seal population, with something like 66,000 of them occupying the beach where penguins attempt to breed. Landslides close to cliff edges have probably also contributed to penguin declines, displacing some and killing others.
But wait a minute… The study included a spell in the 2007/2008 Austral summer, right? According to online sources, the field station at Bouvetøya had been destroyed in 2007, likely as a result of an earthquake followed by a severe winter storm. I contacted Kit Kovacs of the Norwegian Polar Institute in Tromsø, and corresponding author on the penguin paper. She explained to me that the crew operated under difference circumstances in 2008. “The tent was a super insulated Canadian product that made a fine home for the crew,” she wrote. “We have a wonderful new station now, so the tent is old news and fading from memory.” One member was bitten by a fur seal that year, but those are the sorts of risks that a field biologist has to deal with. The scar probably serves as a reminder of a productive summer spent in a very, very remote spot.
Niemandt, C., et al. 2016. Chinstrap and Macaroni penguin diet and demography at Nyrøysa, Bouvetøya. Antarctic Science 28:91-100.
Photo credits: Chinstrap Penguins, photo by Pete Morris – www.birdquest-tours.com; Macaroni Penguins - knowledgebase.lookseek.com
Week 7 – 20 August 2016:
When Snakes Control the Show
Why might a female Acadian Flycatcher in Connecticut choose to produce three eggs instead of four? Why would a Eurasian Blackbird overwintering in northern Africa choose to fly all the way to Iceland to breed instead of staying put? Why would a Shiny Cowbird in Argentina choose to lay its eggs in the nest of a Rufous-collared Sparrow instead of raising its own chicks? The world of birds is full of odd life history riddles that all seem to have the same solution. By behaving one way instead of another, an individual is attempting to maximize its lifetime reproductive success. As with anatomical and physiological characteristics, many behaviours are shaped by natural selection.
So why, then, do Japanese Scops-owls and Ryukyu Scops-owls, birds of roughly the same size and shape, living in the same place at the same time, choose to breed more than a month apart?
The question was recently tackled by Masahiro Toyama and colleagues from Hokkaido University and the Kyushu Research Centre. Their study sites were the Kunigami Forest Park and Yona Experimental Forest on Okinawa Island, Japan. One of an island chain, and part of Japan’s southern-most prefecture, Okinawa and its forests are subtropical, without the very large seasonal differences in climate found in temperate regions.
Their subjects were two species of scops-owl, a group of about 70 species, known for their small size and spectacularly complex classification. Both the Japanese and Ryukyu scops-owls are comparatively small, between 20 and 25 cm (8 to 10 inches) tall, and I would probably have trouble telling them apart even if I had one in each hand and a field guide open between them.
Even so, these birds are, most certainly different, and the timing of their breeding seasons was sufficiently dissimilar to attract attention. Toyama et al. utilized cameras and nest boxes to look into the factors that might have shaped this difference in timing.
They found that mean egg-laying date of the Japanese Scops-owl was 17 March, with young departing their nests between early May and early June. The mean egg-laying date of Ryukyu Scops-owls was 8 May, and young did not leave the nest until late June to mid-July. Curiously, while Ryukyu Scops-owls averaged 2.76 eggs per nest, and the Japanese Scops-owls averaged just 2.29 eggs per nest, there was no difference between the species in the mean number of young that survived to leave the nest.
How did Toyama and his colleagues resolve the puzzle? The Ryukyu Scops-owl is a dietary specialist, feeding mostly on bush-crickets. These birds can only breed later in the Spring when their insect food is abundant. Unfortunately, this is the time of year when it is warm enough for the principal predator of eggs and chicks, the Ryukyu odd-tooth snake, to be active. The Japanese Scops-owl is able to avoid most of the predation of its eggs and chicks by snakes by breeding earlier in the cooler part of Spring. Being a dietary generalist, feeding on insects, centipedes, and small reptiles, birds and mammals, a Japanese Scops-owl can feed its chicks when a Ryukyu Scops-owl couldn’t. Individuals of both species are doing what they can to raise as many chicks as possible.
When I contacted him, Toyama explained that he has been investigating species differences in nest site selection by different scops owls. It seems that the decision to build a nest in one spot instead of another is related to seasonal changes in predation risk. Competition between members of the two owl species may also help to drive their differences in breeding season. I asked whether the Ryukyu odd-tooth snake was a problem for researchers on Okinawa. Toyama reassured me that these snakes are non-venomous.
Toyama, M., N. Kotaka and I. Koizumi. 2015. Breeding times and nest predation rates of sympatric scops owls with different dietary niche breadth. Can. J. Zool. 93:841-847.
Photo credits: Scops Owl stamp, designed by Israeli artist Miriam Karol (1926-1994), Israel, 1963 - au.pinterest.com; photograph of a Ryukyu Scops owl – shortwing.co.uk
Week 6 – 14 August 2016:
Europe’s Most Endangered Songbird?
Without taxonomy, there is no biological conservation. It really is that simple. We don’t enact laws to protect creatures, or gather funds for projects to help restore their numbers, until we know exactly what we are dealing with.
And therein lies the problem. Birds are the best-studied wildlife group on the planet, yet we still don’t fully understand who is related to whom; the classification of birds is in a constant state of flux. It is one of the key goals of avian biology to understand how one group of birds is related to another, and each new finding is likely to result in a change in the system of classification.
Consider the Blue Chaffinch. According to the Avibase website (http://avibase.bsc-eoc.org), this resident of two of the Canary Islands off the northwest coast of Africa is also known by the common name Teydefinch. Its two-part scientific name is Fringilla teydea, first applied to the species in 1841 by biologists Webb, Berthelot and Moquin-Tandon. Fringilla is Latin for “finch,” and teydea is a reference to Mount Teyde on the island of Tenerife.
So far so good?
In 1905, the German ornithologist Ernst Hartest subdivided the Blue Chaffinch into two subspecies on the basis of differences in size and plumage. To the birds on the island of Tenerife, he added a third component to the name, making it Fringillla teydea teydea. Members of the second subspecies, found on the island of Gran Canaria, were to be known by the three-part name Fringilla teydea polatzeki. One species – two subspecies.
Still with me?
According to the IUCN’s Red List of Threatened Species website (http://iucnredlist.org), the Blue Chaffinch should be considered “near-threatened.” The IUCN points out that the subspecies living on Gran Canaria is doing very poorly, but when combined with birds on Tenerife, the species is secure and possibly increasing in size. Laws that protect species do not necessarily apply to threatened subspecies. Consequently, the Gran Canaria form of the Blue Chaffinch might soon be lost.
Enter Swedish researcher George Sangster and four of his colleagues from the Canary Islands, the Netherlands, Portugal and the United States. For the first time since 1905, Sangster et al. reconsidered the taxonomy of Blue Chaffinches. Recognizing the value of utilizing multiple lines of evidence, they first considered differences in the plumage of the two chaffinch groups, studying stuffed specimens in museum collections. The group then recorded the calls and songs of Blue Chaffinches in the wild, and accessed archived recordings, to assess differences between the groups. They also conducted playback experiments in which songs of both subspecies were broadcast to Blue Chaffinches on Gran Canaria to determine whether they could discriminate between songs from their own island and those from Tenerife.
When combined with earlier studies of genetic differences between the groups, Sangster and his colleagues felt that differences in plumage, songs, calls, and responses to playback all pointed to the conclusion that Blue Chaffinches on Gran Canaria and Tenerife are not just subspecies, but rather are different species. Already different, they are likely to continue to diverge in the future. Given that the Gran Canaria form has a very small range (less than 20 km2; 8 square miles) and population (something like 120 individuals), and that the population has been in a long-term decline, Sangster et al. felt that Fringilla polatzeki (a full species) should be considered critically endangered and in need of immediate assistance.
When I contacted him, Sangster responded with great enthusiasm. He even supplied the photograph that accompanies this piece. He explained that he and his colleagues had known for more than a decade that the Gran Canaria population of the Blue Chaffinch was something unique and special, but waited to publish their findings until all of the necessary evidence had been collected, and that local ornithologists were in agreement with the change in status. They were also concerned that an influx of birdwatchers might damage the last bit of habitat available to the birds.
Felipe Rodríguez-Godoy added to the story, explaining that a captive breeding program for the Gran Canaria Blue Chaffinch has been in place for several years. To date 80 juvenile birds, products of the breeding program have been released. The population has been growing very slowly, and so a translocation program has been added. Some of these birds were fitted with radio transmitters so that their progress might be monitored. It will be a couple of years before the effectiveness of the translocation is known.
It is through efforts like these that the future of Europe’s most endangered songbird now looks a bit brighter.
Sangster, G., F. Rodríguez-Godoy, C. S. Roselaar, M. S. Robb and J. A. Luksenburg. 2016. Integrative taxonomy reveals Europe’s rarest songbird species the Gran Canaria Blue Chaffinch Fringuilla polatzeki. J. Avian Biol. 47:159-166.
Photocredits: photograph of a male Blue Chaffinch by Steve Kitchen, from http://birdingaxarquia2.blogspot.com.au/2016/03/the-blue-chaffinch.html; painting of Blue Chaffinches from http://www.avibirds.com/html/Blue_Chaffinch.html
Week 5 – 07 August 2016:
Perhaps We Should Begin Again
It is easy to be captivated by the extraordinary efforts of some birds in constructing their nests. Several days are required for a pair of Cliff Swallows to gather the 1000 mud pellets necessary to build their gourd-shaped nest under a bridge. A male Australian Brush-turkey will continue to maintain and modify his mound of rotting vegetation week after week, for as long as there are eggs to be incubated within it. And if nests can be so very costly to construct, why are members of some species willing to abandon a nest before ever using it?
Christa Benkmann of Deakin University in Australia and Kathy Martin of the University of British Columbia in Canada addressed the question of nest abandonment in Grey Fantails, tiny, insect-eating songbirds native to Australia and New Guinea. Their nests are composed of dry grass and bark, enclosed within a layer of spider webs, with an open top and a stem hanging from the bottom. Fully 71% of nests appear to be abandoned before being used, and a few birds might build as many as seven nests in a breeding season. What is the point of constructing a nest just to abandon it?
Benkmann and Martin examined four possible explanations by monitoring the construction of 194 Grey Fantail nests in the 2012/2013 breeding season.
1. Could it be that what we have interpreted as abandonment was actually “cryptic predation?” Perhaps predators are sneaking in and consuming the eggs before the nest was noticed by researchers. This doesn’t seem to be the case in Grey Fantails, as most nests are abandoned before being completed.
2. Perhaps nests are abandoned after being damaged by extreme weather. Possible, but not likely, as Beckmann and Martin found that only 9% of abandonments occurred within 24 hours of bad weather.
3. Could it be that Grey Fantails are constructing decoy nests in order to dissuade potential predators? If a predator investigated one nest after another but didn’t find anything to eat, perhaps it would abandon its search for nests of that sort. This doesn’t seem likely in the case of fantails as some individuals were seen to dismantle abandoned nests, and reuse old nesting material in the construction of new nests.
4. Only one explanation was consistent with the observations of Beckmann and Martin. It seems that Grey Fantail nests that were under construction, but proved to be poorly concealed from predators, were abandoned. New nests were then constructed in more secure circumstances. Bird predators, particularly Pied and Grey currawongs were observed preying on both eggs and nestlings. As visual predators, superior nest concealment is probably essential to avoiding them.
But what actually cues the fantails, telling them that their current nest might not be in the right place? When contacted, Beckman responded that for daytime predators like currawongs, seeing them near the nest is probably enough to cause fantails to abandon the attempt. “I think mammalian predators might also find the nests after dark – maybe they leave scent cues the birds can use?” Beckman is planning to continue her work in this area, using cameras to identify predators at the nest. Very little is known about nest predators in Australia.
Beckman, C. and K. Martin. 2016. Testing hypotheses about the function of repeated nest abandonment as a life history in a passerine bird. Ibis 158:335-342.
Photo credits: incubating grey fantail - strathbogierangesnatureview.wordpress.com; stamp – www.birdtheme.org
Week 4 – 31 July 2016:
Yawn
Yawning is an incredible behaviour. We all do it, but no one can say definitively why we do it. Yawning generally happens more frequently when we are tired or bored, but that doesn’t say much about its function. Some researchers have suggested that yawning is a way of cooling the brain, or that it arouses the brain’s cerebral cortex. Whatever the function, it must be pretty important because lots of animals do it, from jewel fish to walruses.
Are you yawning yet? If so, then you are engaged in contagious yawning, the result of seeing others yawn, or even thinking about yawning. Contagious yawning is a far less common behaviour, and had been demonstrated convincingly for just a handful of mammal species.
Recently Andre Gallup and colleagues in the Psychology Department at the State University of New York in Oneonta investigated contagious yawning in a group of captive budgerigars. Given that budgies form long-term pair bonds, and live in coordinated flocks year-round, they seemed like a good test subject. So… can budgies cause each other to yawn?
Gallup et al. observed paired birds in adjacent cages. In some cases, members of the pair could see each other, and in other cases they were visually isolated by an opaque barrier. After 32 hours of observation, and 131 recorded yawns, it was found that a budgie was more likely to yawn if it had seen its companion do the same. It didn’t seem to matter whether the subject was familiar with the bird in the next cage, or if it was a stranger. Budgies were then shown video footage of 150 yawns. This resulted in a modest by significant increased the frequency of yawning.
A link has been suggested between contagious yawning and the ability to empathize with plight of others. If you are a contagious yawner, it may be that you are a more empathetic person. Gallup et al. suggested that budgerigars might serve as a good model for the study of empathy.
When contacted, Andrew explained that his group was continuing their studies of budgie social behaviour, and was interested in a neurochemical basis for their their contagious yawning.
I asked whether bird yawning caused yawns among members of the Bird Yawning Lab. He said: “I did find myself yawning quite frequently when I first began to study yawning, but over the years I have become habituated to yawning stimuli (at least when it comes to research).”
Gallup, A. C. et al. 2015. Experimental evidence of contagious yawning in budgerigars (Melopsittacus undulatus). Anim. Cogn. 18:1051-1058.
Photo credits: budgies – Talkbudgies.com; Australian Post budgerigar stamp – stampboards.com
Week 3 – 24 July 2016:
Eagles and Snakes
There are many reasons why birds are so appealing. Birds are reasonably large, and active during the day, making them easy to spot. Most have exquisite plumage, exhibit interesting behaviours, and are capable of flight which captures the imagination. We share our immediate world with many types of birds. As a consequence, birds are better-known that any other group of plants or animals, and lessons learned about birds are likely to apply to other species. And so our studies of avian biology continue.
In a recent paper, Ezra Hadad of the Israel Nature and Parks Authority, and Gilad Weil and Motti Charter of the University of Haifa pointed out that birds, particularly large species like hawks and eagles, have a special role to play in conservation biology. If we protect birds with large home ranges, they explained, then we protect all of the other plants and animals within that space. However, a thorough understanding of the natural history of a species, particularly the factors that limit its abundance, is necessary for the formulation of effective plans for preservation.
Over three breeding seasons, Hadad and his colleagues studied Short-toed Snake-eagles over a 1,175 km2 (453 mi2) section of the Judea region of Israel. (For perspective, this is nearly one-twentieth of Israel’s entire area). The study area was surveyed at least once a week through each breeding season (March to August). Eagle nesting sites were located by watching for courtship and territorial behaviours, and for adults flying while carrying prey.
As many as 109 breeding pairs of Short-toed Snake-eagles were monitored each year, and the reproductive success of these pairs was as high as 98% – an exceptional result. On average nests were situated about 1 km (0.6 mi) from their nearest neighbours, although two exceptional nests were discovered just 58 metres (190 feet) apart. Perhaps most importantly, regions of natural habitat had a significantly higher density of eagle nests than agricultural areas or human settlements. This may be related to availability of the eagle’s preferred food; as its name implies, this species is particularly fond of snakes. The loss of trees in which the eagles nest is also a concern.
When contacted, Motti Charter explained that Israeli laws against hunting are effective in protecting Short-toed Snake-eagles, but “since the country is small with many borders, birds of prey frequently fly over the borders to hunt and are at risk.” Charter provided me with other fascinating aspects of the natural history of this peculiar species. He said that some of the snakes in Israel are venomous, but that the eagles seem to be immune to the venom. Even so, the snake wins the battle on some occasions, particularly if the eagle attempts to capture a constrictor by the head or the tail, rather than in the middle of the body. As the name suggests, the eagle has short toes, which aids in capturing snakes.
Charter also sent me a link to a camera providing live coverage of a pair of Short-toed Snake-eagles at their nest, which you may wish to see. http://www.birds.org.il/he/tracking-methods.aspx?camId=22.
With a population of 8.4 million, Israel has about as many residents as the American state of Virginia, living on just one-fifth of Virginia’s land mass. According to the Israel Central Bureau of Statistics, the nation’s population is likely to grow to between 10.0 million and 12.8 million by the year 2035. Throughout Israel and other parts of the Middle East, Hadad and his colleagues explained, natural habitat is being encroached upon by human settlements and agricultural land. Let’s hope that development plans are made wisely, so as to preserve the region’s precious biological diversity.
Hadad, E., Weil, G., and M. Charter. 2015. The importance of natural habitats as Short-toed Eagle (Circaetus gallicus) breeding sites. Avian Biology Research 8:160-166.
Photo credits: Short-toed Snake-eagle stamp from Azerbaijan – Colnet.com; Short-toed Snake-eagle print from the Natural History of European Birds, published in Zurich in 1964 – www.etsy.com
Week 2 – 17 July 2016:
The Flexibility of Bird Behaviour
Birds are models of marital bliss. Given that there are something like 10,000 species of birds, it is not surprising that there are plenty of exceptions, but the most common avian breeding system, by far, is monogamy. The specific roles of mother and father offer differ, but in most cases both parents contribute to raising the young. One Mommy, one Daddy, and lots of happy chicks.
If that were the end of the story, it would be a rather boring one, and birds are never boring. The subtle variations on the theme of social monogamy in birds are myriad. One of these variations is interspecific brood parasitism in which females of one species lay their eggs in the nests of birds of another species, leaving the cost of raising their offspring to others. Most bird enthusiasts will be familiar with interspecific brood parasites including Brown-headed Cowbirds in North America, Common Cuckoos in Europe, and Pacific Koel in Australia. Each takes advantage of a wide range of hosts.
Less well known is the phenomenon of conspecific brood parasitism (CBP). In this situation, females lay eggs in the nest of other members of the same species. Birds can be sneaky, and simply detecting CBP is a challenge, so that researchers often rely on genetic mismatches between chicks and their mothers to show that it has happened.
The benefit to the parasite is easy to imagine. A lot of eggs means a lot of hungry chicks. By leaving the eggs with someone else, a female might greatly increase her lifetime reproductive output. In most cases of CBP studied to date, the costs of parasitism to the host have been reasonably small. Ducks and geese provide comparatively little direct care to their young, and a one more chick is unlikely to be costly.
The situation is very different in birds like the Prothonotary Warbler. Breeding in woodlands in the southeastern United States, this warbler nests in natural cavities and nest boxes. As with other songbirds, the young are completely dependent on their parents for care.
Anna Tucker and her colleagues at Virginia Commonwealth University and the College of William and Mary studied conspecific brood parasitism in a population of Prothonotary Warblers near Richmond, Virginia. Many of these birds utilized nest boxes provided by the researchers. Anticipating that CBP would be costly to the individuals whose nests received eggs from other individuals, they compared the reproductive success of hosts to those birds that were not parasitized. Study animals received leg bands for future identification, and had blood samples taken for genetic analysis.
Of 233 broods studied, sixty had one or more chicks that were not the offspring of their social mother. Overall, more than twelve percent of nestlings were parasitic. Not surprisingly, females that had been parasitized by another warbler had more chicks leave the nest, but fledged fewer of their own young than did females who were not parasitized. Being parasitized was costly. Tucker and her coworkers found evidence that at least some of the parasitic females were “floaters,” individuals that had been unable to secure their own nest sites, and so resorted to dumping eggs in the nests of others.
When I contacted her, Tucker explained that one of the most interesting aspects of the ongoing study of Prothonotary Warblers was the effects that the addition of nest boxes has had on the population. “These boxes were first established about 25 years ago to support a then-declining population.” The population has now grown almost to the point of overcrowding. This has probably created the conditions for females to dump eggs. “This flexibility on the part of the females laying parasitic eggs has also reshaped the way I think about avian breeding behaviors,” she wrote. Breeding systems in birds are much more complex that we have considered them to be in the past.
Tucker, A. M., et al. 2016. Opportunistic conspecific brood parasitism in a box-nesting population of Prothonotary Warblers (Protonotaria citrea). Auk 133:29
Photocredits: Prothonotary Warbler stamp, www.canadapost.com; singing Prothonotary Warbler, www.carolinabirdclub.org
Week 1 – 10 July 2016:
Manna from Australia
It seems that Heaven is not the only source of manna. When wounded, some Australian gum trees respond by releasing a syrupy material at the site of the injury. The white gum tree, also known as the manna gum, Eucalyptus viminalis, reacts to damage to its leaves and stems by releasing a sugary fluid that soon crystalizes into a mass that can be eaten by a range of arthropods and a few types of birds. Most of these manna-eating animals are probably opportunists, taking advantage of the yummy treat when they stumble across it. According to recent research by Samuel Case and Amanda Edworthy of the Australian National University in Canberra, Forty-spotted Pardalotes, Pardalotus quadragintus, are an exception. They don’t just harvest manna; these pardalotes mine it.
At one time, the Forty-spotted Pardalote could be found throughout eastern portions of the Australian state of Tasmania. Those days are gone. The bird is a habitat specialist, relying on forests with manna gums. With the clearing of those woodlots, combined with recent drought, the pardalote’s range has contracted to the point where it is now considered an endangered species. If we are to fully understand the decline of the Forty-spotted Pardalote, and perhaps assist in its recovery, we need to understand the relationship between the bird and its habitat.
While studying the breeding biology of Forty-spotted Pardalotes, Case and Edworthy observed these birds using a peculiar notch toward the end of their beaks to make slits in the leaf stalks of mana gum trees. This caused the plants to produce sugary manna. How common is this behaviour, and how important is manna to the lives of the birds?
Between August 2014 and January of 2015, Case and Edworthy made observations of pardalotes in and around nesting sites. They supplemented their direct observations with recordings by digital video cameras placed near nest entrances, capturing images of the food brought to nestlings by their parents. The researchers also became adept at damaging lead stalks artificially to determine what portion of the gum trees produced manna.
More than 80% of the diet of Forty-spotted Pardalote nestlings was manna, supplemented by a few flies and spiders. It has yet to be determined whether manna is rich in amino acids, or if a few arthropods are necessary for a well-balanced diet for the nestlings. Given that manna is chosen as a food for nestlings more often than invertebrates, Case and Edworthy expect that the plant’s exudate provides substantial nutrients.
The findings of Case and Edworthy have profound implications for conservation of the endangered Forty-spotted Pardalote. If captive breeding should ever become necessary, the manna gum will surely be a feature of their enclosure. “All white gum trees may not produce manna equally,” Case explained to me, “and a better understanding of manna production could help to conserve habitat for these endangered birds.”
Are you like me? Are you aching to know what manna tastes like? According to Case, manna “tastes similar to a breath mint.” Now you know.
Case, S. B., and A. B. Edworthy. 2016. First report of ‘mining’ as a feeding behaviour among Australian manna-feeding birds. Ibis 158:407-415.
Photo credits: Forty-spotted Pardalote stamp, www.birdtheme.org; Forty-spotted Pardalote in a manna gum, www.nrmsouth.org.au/forty-spotted-pardalote/

















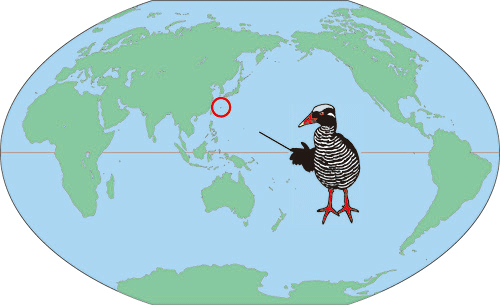



























































































































































































Pingback: A Traveller’s Guide: Auklets – Christy N. Wails